Quinine hemisulfate hydrate
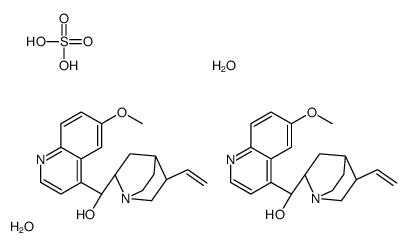
Quinine hemisulfate hydrate structure
|
Common Name | Quinine hemisulfate hydrate | ||
|---|---|---|---|---|
| CAS Number | 207671-44-1 | Molecular Weight | 782.94300 | |
| Density | N/A | Boiling Point | N/A | |
| Molecular Formula | C20H24N2O2.1/2H2O4S.H2O | Melting Point | N/A | |
| MSDS | Chinese USA | Flash Point | N/A | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of Quinine hemisulfate hydrateQuinine hemisulfate hydrate is an alkaloid derived from the bark of the cinchona tree, acts as an anti-malaria agent. Quinine hemisulfate hydrate is a potassium channel inhibitor that inhibits WT mouse Slo3 (KCa5.1) channel currents evoked by voltage pulses to +100 mV with an IC50 of 169 μM[1][2]. |
| Name | Hydrogen sulfate-(8α,9R)-6'-methoxycinchonan-9-ol hydrate (2:1: 2:2) |
|---|---|
| Synonym | More Synonyms |
| Description | Quinine hemisulfate hydrate is an alkaloid derived from the bark of the cinchona tree, acts as an anti-malaria agent. Quinine hemisulfate hydrate is a potassium channel inhibitor that inhibits WT mouse Slo3 (KCa5.1) channel currents evoked by voltage pulses to +100 mV with an IC50 of 169 μM[1][2]. |
|---|---|
| Related Catalog | |
| Target |
IC50: Parasite[1], 169 μM (mSlo3)[2] |
| References |
| Molecular Formula | C20H24N2O2.1/2H2O4S.H2O |
|---|---|
| Molecular Weight | 782.94300 |
| Exact Mass | 782.35600 |
| PSA | 192.62000 |
| LogP | 6.52160 |
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H315-H319-H335 |
| Precautionary Statements | P261-P305 + P351 + P338 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xi |
| RIDADR | NONH for all modes of transport |
|
Interaction of quinine sulfate with anionic micelles of sodium dodecylsulfate: A time-resolved fluorescence spectroscopy at different pH.
Spectrochim. Acta. A. Mol. Biomol. Spectrosc. 148 , 49-59, (2015) Photophysical behavior and rotational relaxation dynamics of quinine sulfate (QS) in anionic surfactant, sodium dodecylsulfate (SDS) at different pH have been studied using steady state and time resol... |
|
|
The uptake of trehalose glycolipids by macrophages is independent of Mincle.
ChemBioChem. 16(4) , 683-93, (2015) Trehalose glycolipids play an important role in the pathogenesis of Mycobacterium tuberculosis and are used as adjuvants for vaccines; however, much still remains unanswered about the mechanisms throu... |
|
|
In vivo antimalarial activity and mechanisms of action of 4-nerolidylcatechol derivatives.
Antimicrob. Agents Chemother. 59 , 3271-80, (2015) 4-Nerolidylcatechol (1) is an abundant antiplasmodial metabolite that is isolated from Piper peltatum roots. O-Acylation or O-alkylation of compound 1 provides derivatives exhibiting improved stabilit... |
| Quinine hemisulfate |
| Quinine sulfate |