Moxifloxacin HCl
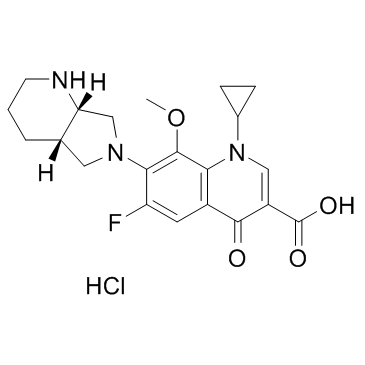
Moxifloxacin HCl structure
|
Common Name | Moxifloxacin HCl | ||
|---|---|---|---|---|
| CAS Number | 186826-86-8 | Molecular Weight | 437.892 | |
| Density | N/A | Boiling Point | 636.4ºC at 760 mmHg | |
| Molecular Formula | C21H25ClFN3O4 | Melting Point | N/A | |
| MSDS | Chinese USA | Flash Point | 338.7ºC | |
Use of Moxifloxacin HClMoxifloxacin (Hydrochloride) is a synthetic fluoroquinolone antibiotic agent.Target: AntibacterialMoxifloxacin is an extended-spectrum fluoroquinolone which has improved coverage against gram-positive cocci and atypical pathogens compared with older fluoroquinolone agents, while retaining good activity against gram-negative bacteria. The antibacterial spectrum of moxifloxacin includes all major upper and lower respiratory tract pathogens; it is one of the most active fluoroquinolones against pneumococci, including penicillin- and macrolide-resistant strains [1]. Moxifloxacin has limited phototoxic potential. In clinical trials, moxifloxacin had clinical success rates of 88-97% and bacteriologic eradication rates of 90-97%. Moxifloxacin is a safe and effective antimicrobial that will be useful for treating acute sinusitis, acute bacterial exacerbations of chronic bronchitis, and community-acquired pneumonia [2]. Moxifloxacin possibly stimulates lipid peroxidation and enhances phagocytosis, as depicted by MDA production and survival prolongation, without being toxic as depicted by white blood cell count [3]. Clinical indications: Abdominal abscess; Acute bronchitis; Acute sinusitis; Bacterial infectionToxicity: Symptoms of overdose include CNS and gastrointestinal effects such as decreased activity, somnolence, tremor, convulsions, vomiting, and diarrhea. The minimal lethal intravenous dose in mice and rats is 100 mg/kg. |
| Name | moxifloxacin hydrochloride |
|---|---|
| Synonym | More Synonyms |
| Description | Moxifloxacin (Hydrochloride) is a synthetic fluoroquinolone antibiotic agent.Target: AntibacterialMoxifloxacin is an extended-spectrum fluoroquinolone which has improved coverage against gram-positive cocci and atypical pathogens compared with older fluoroquinolone agents, while retaining good activity against gram-negative bacteria. The antibacterial spectrum of moxifloxacin includes all major upper and lower respiratory tract pathogens; it is one of the most active fluoroquinolones against pneumococci, including penicillin- and macrolide-resistant strains [1]. Moxifloxacin has limited phototoxic potential. In clinical trials, moxifloxacin had clinical success rates of 88-97% and bacteriologic eradication rates of 90-97%. Moxifloxacin is a safe and effective antimicrobial that will be useful for treating acute sinusitis, acute bacterial exacerbations of chronic bronchitis, and community-acquired pneumonia [2]. Moxifloxacin possibly stimulates lipid peroxidation and enhances phagocytosis, as depicted by MDA production and survival prolongation, without being toxic as depicted by white blood cell count [3]. Clinical indications: Abdominal abscess; Acute bronchitis; Acute sinusitis; Bacterial infectionToxicity: Symptoms of overdose include CNS and gastrointestinal effects such as decreased activity, somnolence, tremor, convulsions, vomiting, and diarrhea. The minimal lethal intravenous dose in mice and rats is 100 mg/kg. |
|---|---|
| Related Catalog | |
| References |
| Boiling Point | 636.4ºC at 760 mmHg |
|---|---|
| Molecular Formula | C21H25ClFN3O4 |
| Molecular Weight | 437.892 |
| Flash Point | 338.7ºC |
| Exact Mass | 437.151764 |
| PSA | 83.80000 |
| LogP | 3.56630 |
| Vapour Pressure | 4.56E-17mmHg at 25°C |
| Storage condition | 2-8°C |
| Hazard Codes | Xi |
|---|---|
| RIDADR | NONH for all modes of transport |
| RTECS | VB1983750 |
| HS Code | 2933499090 |
|
~74% 
Moxifloxacin HCl CAS#:186826-86-8 |
| Literature: CIPLA LIMITED; CURTIS, Philip, Anthony Patent: WO2008/59223 A2, 2008 ; Location in patent: Page/Page column 15-16 ; |
|
~% 
Moxifloxacin HCl CAS#:186826-86-8 |
| Literature: WO2010/52726 A1, ; Page/Page column 5; 6 ; |
|
~73% 
Moxifloxacin HCl CAS#:186826-86-8 |
| Literature: CIPLA LIMITED; CURTIS, Philip, Anthony Patent: WO2008/59223 A2, 2008 ; Location in patent: Page/Page column 13-14 ; |
|
~% 
Moxifloxacin HCl CAS#:186826-86-8 |
| Literature: WO2007/10555 A2, ; Page/Page column 15 ; |
|
~% 
Moxifloxacin HCl CAS#:186826-86-8 |
| Literature: EP2551268 A1, ; |
|
~% 
Moxifloxacin HCl CAS#:186826-86-8 |
| Literature: EP2551268 A1, ; |
|
~% 
Moxifloxacin HCl CAS#:186826-86-8 |
| Literature: Spectrochimica Acta - Part A: Molecular and Biomolecular Spectroscopy, , vol. 121, p. 254 - 258 |
|
~% 
Moxifloxacin HCl CAS#:186826-86-8 |
| Literature: Spectrochimica Acta - Part A: Molecular and Biomolecular Spectroscopy, , vol. 121, p. 254 - 258 |
|
~% 
Moxifloxacin HCl CAS#:186826-86-8 |
| Literature: Spectrochimica Acta - Part A: Molecular and Biomolecular Spectroscopy, , vol. 121, p. 254 - 258 |
| HS Code | 2933990090 |
|---|---|
| Summary | 2933990090. heterocyclic compounds with nitrogen hetero-atom(s) only. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Simple and accurate quantitative analysis of 20 anti-tuberculosis drugs in human plasma using liquid chromatography-electrospray ionization-tandem mass spectrometry.
J. Pharm. Biomed. Anal. 102 , 9-16, (2014) A simple and accurate liquid chromatography (LC)-tandem mass spectrometry (MS/MS) method for the quantitation of 20 anti-tuberculosis (anti-TB) drugs in human plasma, was developed as a tool for thera... |
|
|
Exogenous dopamine induces dehydroepiandrosterone sulfotransferase (rSULT2A1) in rat liver and changes the pharmacokinetic profile of moxifloxacin in rats.
Drug Metab. Pharmacokinet. 30(1) , 97-104, (2015) Dehydroepiandrosterone sulfotransferase (SULT2A1) plays an important role in the detoxification of hydroxyl-containing xenobitotics and in the regulation of the biological activities of hydroxysteroid... |
|
|
Effect of norfloxacin and moxifloxacin on melanin synthesis and antioxidant enzymes activity in normal human melanocytes.
Mol. Cell Biochem. 401(1-2) , 107-14, (2015) Fluoroquinolone antibiotics provide broad-spectrum coverage for a number of infectious diseases, including respiratory as well as urinary tract infections. One of the important adverse effects of thes... |
| 1-cyclopropyl-6-fluoro-1,4-dihydro-8-methoxy-7-[(4aS,7aS)-octahydro-6H-pyrrolo[3,4-b]pyridine-6-yl]-4-oxo-3-quinolinecarboxylic acid hydrochloride |
| BAY-12-8039 |
| Moxifloxacinehydrochloride |
| 3-quinolinecarboxylic acid, 1-cyclopropyl-6-fluoro-1,4-dihydro-8-methoxy-7-[(4aS,7aS)-octahydro-6H-pyrrolo[3,4-b]pyridin-6-yl]-4-oxo-, monohydrochloride |
| Moxifloxacin |
| 1-Cyclopropyl-6-fluoro-8-methoxy-7-[(4aS,7aS)-octahydro-6H-pyrrolo[3,4-b]pyridin-6-yl]-4-oxo-1,4-dihydroquinoline-3-carboxylic acid hydrochloride (1:1) |
| Moxiflaxacin HCL |
| Avelox,Avalox |
| Moxifloxacin Hydrochloride (200 mg) |
| Vigamox |
| 1-cyclopropyl-6-fluoro-8-methoxy-7-[(4aS,7aS)-octahydropyrrolo[3,4-b]pyridin-6-yl]-4-oxo-1,4-dihydroquinoline-3-carboxylic acid hydrochloride |
| acide 1-cyclopropyl-6-fluoro-8-méthoxy-7-[(4aS,7aS)-octahydro-6H-pyrrolo[3,4-b]pyridin-6-yl]-4-oxo-1,4-dihydroquinoléine-3-carboxylique chlorhydrate |
| MoxifloxacineHCl |
| Octegra |
| 1-Cyclopropyl-6-fluor-8-methoxy-7-[(4aS,7aS)-octahydro-6H-pyrrolo[3,4-b]pyridin-6-yl]-4-oxo-1,4-dihydrochinolin-3-carbonsäurehydrochlorid |
| Izilox |
| 1-cyclopropyl-7-(S,S)-2,8-diazabicyclo(4.3.0)-non-8-yl-6-fluoro-8-methoxy-1,4-dihydro-4-oxo-3-quinoline carboxylic acid hydrochloride |
| 1-cyclopropyl-6-fluoro-8-methoxy-7-[(4aS,7aS)-octahydro-6H-pyrrolo[3,4-b]pyridin-6-yl]-4-oxo-1,4-dihydroquinoline-3-carboxylic acid hydrochloride |
| MOXIFLOXACIN HCL |
| Moxiflocacin hydrochloride |
| 1-Cyclopropyl-6-fluoro-8-methoxy-7-((4aS,7aS)-octahydro-6H-pyrrolo[3,4-b]pyridin-6-yl)-4-oxo-1,4-dihydro-3-quinolinecarboxylic Acid Hydrochloride |
| 3-Quinolinecarboxylic acid, 1-cyclopropyl-6-fluoro-1,4-dihydro-8-methoxy-7-[(4aS,7aS)-octahydro-6H-pyrrolo[3,4-b]pyridin-6-yl]-4-oxo-, hydrochloride (1:1) |
| MFCD00949117 |
| Moxifloxacin hydrochloride |
| (1'S,6'S)-1-cyclopropyl-7-(2,8-diazabicyclo[4.3.0]non-8-yl)-6-fluoro-8-methoxy-4-oxo-(1,4-dihydroquinoline)-3-carboxylic acid hydrochloride salt |
| Actira |
| 1-Cyclopropyl-6-fluoro-8-methoxy-7-[(4aS,7aS)-octahydro-6H-pyrrolo[3,4-b]pyridin-6-yl]-4-oxo-1,4-dihydro-3-quinolinecarboxylic acid hydrochloride (1:1) |
| 1-cyclopropyl-6-fluoro-1,4-dihydro-8-methoxy-7-[(4aS,7aS)-octahydro-6H-pyrrolo-[3,4-b]pyridin-6-yl]-4-oxo-3-quinolinecarboxylic acid hydrochloride |
| Moxifloxacin (Hydrochloride) |
| Proflox |
| Avelox |
| Avalox |
![(S,S)-2,8-Diazabicyclo[4,3,0]nonane structure](https://image.chemsrc.com/caspic/273/151213-40-0.png)


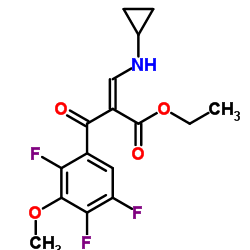
![(s,s)-6-benzyl-octahydro-pyrrolo[3,4-b]pyridine dihydrochloride structure](https://image.chemsrc.com/caspic/138/151213-39-7.png)

