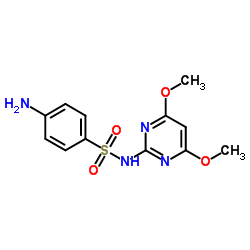
CHEMICAL IDENTIFICATION
-
RTECS NUMBER :
-
DA9495000
-
CHEMICAL NAME :
-
Benzenesulfonamide, 4-amino-N-(4,6-dimethoxy-2-pyrimidinyl)-
-
CAS REGISTRY NUMBER :
-
155-91-9
-
BEILSTEIN REFERENCE NO. :
-
0307123
-
LAST UPDATED :
-
199612
-
DATA ITEMS CITED :
-
7
-
MOLECULAR FORMULA :
-
C12-H14-N4-O4-S
-
MOLECULAR WEIGHT :
-
310.36
-
WISWESSER LINE NOTATION :
-
T6N CNJ BMSWR DZ& DO1 FO1
HEALTH HAZARD DATA
ACUTE TOXICITY DATA
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Unreported
-
SPECIES OBSERVED :
-
Rodent - rat
-
DOSE/DURATION :
-
28 gm/kg/28D-I
-
TOXIC EFFECTS :
-
Liver - hepatitis (hepatocellular necrosis), zonal Blood - normocytic anemia Related to Chronic Data - death
-
REFERENCE :
-
BJPCAL British Journal of Pharmacology and Chemotherapy. (London, UK) V.1-33, 1946-68. For publisher information, see BJPCBM. Volume(issue)/page/year: 2,149,1947 ** REPRODUCTIVE DATA **
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
DOSE :
-
538 mg/kg
-
SEX/DURATION :
-
female 1-22 day(s) after conception lactating female 21 day(s) post-birth
-
TOXIC EFFECTS :
-
Reproductive - Specific Developmental Abnormalities - craniofacial (including nose and tongue) Reproductive - Effects on Newborn - physical
-
REFERENCE :
-
BJPCAL British Journal of Pharmacology and Chemotherapy. (London, UK) V.1-33, 1946-68. For publisher information, see BJPCBM. Volume(issue)/page/year: 23,305,1964
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
DOSE :
-
1 gm/kg
-
SEX/DURATION :
-
female 14 day(s) after conception
-
TOXIC EFFECTS :
-
Reproductive - Specific Developmental Abnormalities - musculoskeletal system Reproductive - Effects on Newborn - stillbirth
-
REFERENCE :
-
BJPCAL British Journal of Pharmacology and Chemotherapy. (London, UK) V.1-33, 1946-68. For publisher information, see BJPCBM. Volume(issue)/page/year: 23,305,1964
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
DOSE :
-
825 mg/kg
-
SEX/DURATION :
-
female 1-22 day(s) after conception lactating female 21 day(s) post-birth
-
TOXIC EFFECTS :
-
Reproductive - Effects on Newborn - weaning or lactation index (e.g., # alive at weaning per # alive at day 4)
-
REFERENCE :
-
BJPCAL British Journal of Pharmacology and Chemotherapy. (London, UK) V.1-33, 1946-68. For publisher information, see BJPCBM. Volume(issue)/page/year: 23,305,1964
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
DOSE :
-
500 mg/kg
-
SEX/DURATION :
-
female 11-20 day(s) after conception
-
TOXIC EFFECTS :
-
Reproductive - Specific Developmental Abnormalities - skin and skin appendages
-
REFERENCE :
-
EXPEAM Experientia. (Birkhaeuser Verlag, POB 133, CH-4010 Basel, Switzerland) V.1- 1945- Volume(issue)/page/year: 21,151,1965
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
DOSE :
-
2520 mg/kg
-
SEX/DURATION :
-
female 1-22 day(s) after conception lactating female 21 day(s) post-birth
-
TOXIC EFFECTS :
-
Reproductive - Effects on Newborn - stillbirth Reproductive - Effects on Newborn - weaning or lactation index (e.g., # alive at weaning per # alive at day 4)
-
REFERENCE :
-
BJPCAL British Journal of Pharmacology and Chemotherapy. (London, UK) V.1-33, 1946-68. For publisher information, see BJPCBM. Volume(issue)/page/year: 23,305,1964
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
DOSE :
-
2520 mg/kg
-
SEX/DURATION :
-
female 1-22 day(s) after conception lactating female 21 day(s) post-birth
-
TOXIC EFFECTS :
-
Reproductive - Specific Developmental Abnormalities - eye/ear Reproductive - Specific Developmental Abnormalities - craniofacial (including nose and tongue) Reproductive - Specific Developmental Abnormalities - musculoskeletal system
-
REFERENCE :
-
BJPCAL British Journal of Pharmacology and Chemotherapy. (London, UK) V.1-33, 1946-68. For publisher information, see BJPCBM. Volume(issue)/page/year: 23,305,1964
|