Sodium tert-pentoxide
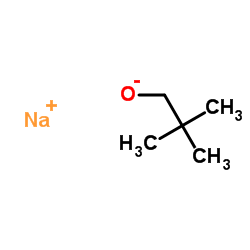
Sodium tert-pentoxide structure
|
Common Name | Sodium tert-pentoxide | ||
|---|---|---|---|---|
| CAS Number | 14593-46-5 | Molecular Weight | 110.130 | |
| Density | 0.904 g/mL at 25 °C | Boiling Point | 113.1ºC at 760 mmHg | |
| Molecular Formula | C5H11NaO | Melting Point | 200°C | |
| MSDS | Chinese USA | Flash Point | -21 °C | |
| Symbol |


GHS02, GHS05 |
Signal Word | Danger | |
| Name | Sodium tert-pentoxide |
|---|---|
| Synonym | More Synonyms |
| Density | 0.904 g/mL at 25 °C |
|---|---|
| Boiling Point | 113.1ºC at 760 mmHg |
| Melting Point | 200°C |
| Molecular Formula | C5H11NaO |
| Molecular Weight | 110.130 |
| Flash Point | -21 °C |
| Exact Mass | 110.070763 |
| PSA | 23.06000 |
| LogP | 1.60550 |
| Vapour Pressure | 19.2mmHg at 25°C |
| InChIKey | CGRKYEALWSRNJS-UHFFFAOYSA-N |
| SMILES | CCC(C)(C)[O-].[Na+] |
| Storage condition | Flammables + water-Freezer (-20°C)e area |
| Symbol |


GHS02, GHS05 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H251-H314 |
| Supplemental HS | Reacts violently with water. |
| Precautionary Statements | P235 + P410-P280-P305 + P351 + P338-P310 |
| Personal Protective Equipment | Eyeshields;Faceshields;full-face particle respirator type N100 (US);Gloves;respirator cartridge type N100 (US);type P1 (EN143) respirator filter;type P3 (EN 143) respirator cartridges |
| Hazard Codes | F:Flammable;C:Corrosive; |
| Risk Phrases | R11;R14;R35 |
| Safety Phrases | S6-S22-S33-S36/37/39-S7/8-S45-S43C-S26-S16 |
| RIDADR | UN 3206 4.2/PG 2 |
| WGK Germany | 1 |
| Packaging Group | II |
| Hazard Class | 4.1 |
| HS Code | 2942000000 |
|
~% 
Sodium tert-pen... CAS#:14593-46-5 |
| Literature: Journal of Organic Chemistry, , vol. 46, # 22 p. 4331 - 4339 |
| Precursor 1 | |
|---|---|
| DownStream 10 | |
| HS Code | 2915900090 |
|---|---|
| Summary | 2915900090 other saturated acyclic monocarboxylic acids and their anhydrides, halides, peroxides and peroxyacids; their halogenated, sulphonated, nitrated or nitrosated derivatives VAT:17.0% Tax rebate rate:9.0% Supervision conditions:AB(certificate of inspection for goods inward,certificate of inspection for goods outward) MFN tariff:5.5% General tariff:30.0% |
|
Heterolysis of Dihydrogen by Silver Alkoxides and Fluorides.
Chemistry 21 , 10160-9, (2015) Alkoxide-bridged disilver cations react with dihydrogen to form hydride-bridged cations, releasing free alcohol. Hydrogenolysis of neutral silver fluorides affords hydride-bridged disilver cations as ... |
| sodium 2-methylpropan-2-olate |
| MFCD00064245 |
| sodium tert-butanolate |
| sodium tertiary-butoxide |
| sodium t-butanolate |
| Na-t-pentoxide |
| sodium tert-amylate |
| sodium tert-butylate |
| tBuONa |
| Sodium-t-amylate |
| 1-Propanol, 2,2-dimethyl-, sodium salt (1:1) |
| sodium t-butoxide |
| potassium tert-butylate |
| sodium-tert-butoxide |
| Sodium 2,2-dimethyl-1-propanolate |
| NaOtBu |
| t-butoxy sodium |
| NaOt-amylate |
| Sodium tert-Pentoxide |
| Na t-amylate |
| EINECS 238-639-4 |
| Sodium 2-Methyl-2-butoxide |
| t-pentylONa |
| Sodium tert-pentyloxide |
| sodium tert-pentylate |
 CAS#:4282-31-9
CAS#:4282-31-9 CAS#:3580-38-9
CAS#:3580-38-9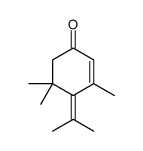 CAS#:4705-09-3
CAS#:4705-09-3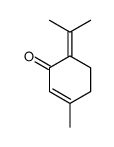 CAS#:491-09-8
CAS#:491-09-8![4-[(6,7-Dimethoxyquinolin-4-yl)oxy]aniline structure](https://image.chemsrc.com/caspic/169/190728-25-7.png) CAS#:190728-25-7
CAS#:190728-25-7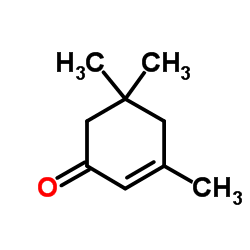 CAS#:78-59-1
CAS#:78-59-1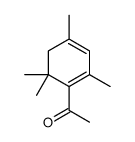 CAS#:13834-80-5
CAS#:13834-80-5 CAS#:67-64-1
CAS#:67-64-1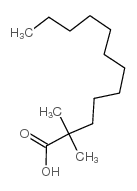 CAS#:2874-73-9
CAS#:2874-73-9 CAS#:59130-75-5
CAS#:59130-75-5
