CNDAC
Modify Date: 2025-08-25 11:24:11
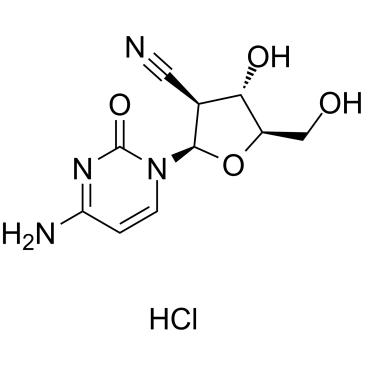
CNDAC structure
|
Common Name | CNDAC | ||
|---|---|---|---|---|
| CAS Number | 135598-68-4 | Molecular Weight | 252.22700 | |
| Density | N/A | Boiling Point | N/A | |
| Molecular Formula | C10H12N4O4 | Melting Point | N/A | |
| MSDS | N/A | Flash Point | N/A | |
Use of CNDACCNDAC is a major metabolite of oral drug sapacitabine, and a nucleoside analog. |
| Name | (2R,3S,4S,5R)-2-(4-amino-2-oxopyrimidin-1-yl)-4-hydroxy-5-(hydroxymethyl)oxolane-3-carbonitrile |
|---|---|
| Synonym | More Synonyms |
| Description | CNDAC is a major metabolite of oral drug sapacitabine, and a nucleoside analog. |
|---|---|
| Related Catalog | |
| In Vitro | CNDAC-induced SSBs can be repaired by the transcription-coupled nucleotide excision repair pathway, whereas lethal DSBs are mainly repaired through homologous recombination. Deficiency in two Rad51 paralogs, Rad51D and XRCC3, greatly sensitize cells to CNDAC. The Rad51D-null cell line is approximately 50-fold more sensitive to CNDAC (IC50=0.006 µM) compared to 51D1.3, the Rad51D-repleted line (IC50=0.32 µM)[1]. CNDAC shows inhibitory activity against HL-60 and THP-1 cells with IC50s of 1.58 µM and 0.84 µM. CNDAC (10 μM) results in a significant drop in cell survival compared to the untreated on days 4, 7, and 14. CNDAC is more effective at reducing viability and inducing apoptosis than ara-C at equivalent concentrations in the THP-1 cell line, which is defined as displaying resistance to ara-C[2]. CNDAC induces DSBs, which are products of replication, rather than a consequence of induction of apoptosis. CNDAC causes DNA damage, and DNA-PK and ATR are dispensable for cell survival. CNDAC exhibits potent activity against human fibroblasts deficient in ATM or transfected with an empty vector, approximately 30-fold more than cells repleted with full-length ATM cDNA, with IC50s of 0.01 μM and 0.3 μM, respectively. CNDAC-induced DNA damage is repaired through the homologous recombination pathway[3]. |
| Cell Assay | 1×106 primary BM and PB cells are treated with 1 μM (low), 10 μM (medium), and 100 μM (high) of ara-C or CNDAC or 0.005 μM (low), 0.05 μM (medium) and 0.5 μM (high) mitoxantrone in 24 well plates at 37°C, 5% CO2, and 100% humidity for 4 days. Appropriate untreated controls are included. Postdrug treatment, both PB and BM non-adherent cells are washed to remove compound, replated on M2-10B4 stromal layers, and reincubated at 37°C, 5% CO2, 100% humidity. Cells are analyzed immediately posttreatment and following 3, 7, and 31 days postdrug removal. |
| References |
| Molecular Formula | C10H12N4O4 |
|---|---|
| Molecular Weight | 252.22700 |
| Exact Mass | 252.08600 |
| PSA | 135.38000 |
| InChIKey | DCYBPMFXJCWXNB-JWIUVKOKSA-N |
| SMILES | N#CC1C(O)C(CO)OC1n1ccc(N)nc1=O |
|
~% 
CNDAC CAS#:135598-68-4 |
| Literature: CYCLACEL LIMITED Patent: WO2009/136162 A1, 2009 ; Location in patent: Page/Page column 19 ; |
|
~% 
CNDAC CAS#:135598-68-4 |
| Literature: Matsuda; Nakajima; Azuma; Tanaka; Sasaki Journal of Medicinal Chemistry, 1991 , vol. 34, # 9 p. 2917 - 2919 |
| Precursor 1 | |
|---|---|
| DownStream 0 | |
| tas-109 |
| cndac |
| unii-00m634hd2v |
![N-{2-Oxo-1-[(6aR,8R,9aR)-2,2,4,4-tetraisopropyl-9-oxotetrahydro-6H-furo[3,2-f][1,3,5,2,4]trioxadisilocin-8-yl]-1,2-dihydro-4-pyrimidinyl}acetamide structure](https://image.chemsrc.com/caspic/091/140859-16-1.png)