Methapyrilene Hydrochloride
Modify Date: 2025-08-25 18:08:12
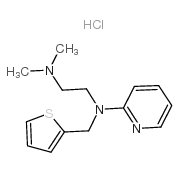
Methapyrilene Hydrochloride structure
|
Common Name | Methapyrilene Hydrochloride | ||
|---|---|---|---|---|
| CAS Number | 135-23-9 | Molecular Weight | 297.84700 | |
| Density | 1.148g/cm3 | Boiling Point | 389.6ºC at 760 mmHg | |
| Molecular Formula | C14H20ClN3S | Melting Point | 162° | |
| MSDS | Chinese USA | Flash Point | 189.4ºC | |
| Symbol |

GHS06 |
Signal Word | Danger | |
Use of Methapyrilene HydrochlorideMethapyrilene (Thenylpyramine) hydrochloride is an orally active H1-receptor antihistamine and an anticholinergic agent of the pyridine chemical class. Methapyrilene hydrochloride has hepatotoxicity and can be used as a hepatotoxin that cause periportal hepatic necrosis in vivo[2] |
| Name | methapyrilene hydrochloride |
|---|---|
| Synonym | More Synonyms |
| Description | Methapyrilene (Thenylpyramine) hydrochloride is an orally active H1-receptor antihistamine and an anticholinergic agent of the pyridine chemical class. Methapyrilene hydrochloride has hepatotoxicity and can be used as a hepatotoxin that cause periportal hepatic necrosis in vivo[2] |
|---|---|
| Related Catalog | |
| Target |
H1 Receptor |
| In Vitro | Methapyrilene hydrochloride (650 μM) results in a down-regulation of TF and up-regulation of FTL, while the level of HMOX1 is not changed. Additionally, the levels of CD44 and SOX9 proteins and the expression of PROM1 (CD133), hepatic stem cell-associated markers are increased[1]. Methapyrilene hydrochloride (650 μM) decreases CYP2E1, CYP3A4, NR1l3, ALB, mRNA expression and increases CD133 expression[1]. |
| In Vivo | Methapyrilene hydrochloride (oral gavage; 40 or 80 mg/kg; 5 days per week; 6 weeks) results in changes in the expression of classic hepatotoxicity-related marker genes and iron homeostasis-related genes, especially a prominent, dose-dependent down-regulation of the transferrin (Tf) gene and an up-regulation of the ferritin, light chain (FTL) gene in rats[1]. Methapyrilene hydrochloride (oral gavage; 150 mg/kg; 3 days) causes periportal liver necrosis at high dosage. Methapyrilene is sufficient to induce liver necrosis, or a subtoxic dose of 50 mg/kg/day[2]. |
| References |
| Density | 1.148g/cm3 |
|---|---|
| Boiling Point | 389.6ºC at 760 mmHg |
| Melting Point | 162° |
| Molecular Formula | C14H20ClN3S |
| Molecular Weight | 297.84700 |
| Flash Point | 189.4ºC |
| Exact Mass | 297.10700 |
| PSA | 47.61000 |
| LogP | 3.51330 |
| Vapour Pressure | 2.82E-06mmHg at 25°C |
| Storage condition | room temp |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Symbol |

GHS06 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H301 |
| Precautionary Statements | P301 + P310 |
| Personal Protective Equipment | Eyeshields;Faceshields;Gloves;type P2 (EN 143) respirator cartridges |
| Hazard Codes | T |
| Risk Phrases | R25 |
| Safety Phrases | S22;S36;S45 |
| RIDADR | UN 2811 6.1/PG 3 |
| WGK Germany | 3 |
| RTECS | UT1750000 |
| Packaging Group | III |
| Hazard Class | 6.1(b) |
| N,N-dimethyl-N'-pyridin-2-yl-N'-(thiophen-2-ylmethyl)ethane-1,2-diamine,hydrochloride |
| Methoxylene |
| EINECS 205-184-8 |
| Methacon |
| Lullamin |
| Histadyl |
| Somnicaps |
| Barhist |
| Coryzol |
| Capathyn |
| MFCD00050665 |
| METHAPYRILENE HYDROCHLORIDE |
| Methapyrilene HCl |