GLYCOL CHITOSAN
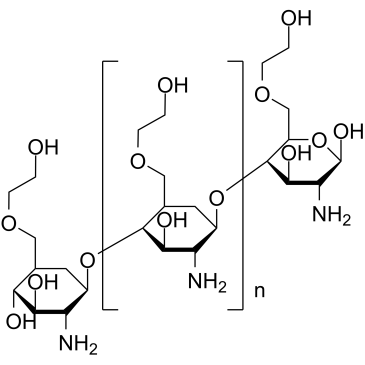
GLYCOL CHITOSAN structure
|
Common Name | GLYCOL CHITOSAN | ||
|---|---|---|---|---|
| CAS Number | 123938-86-3 | Molecular Weight | N/A | |
| Density | N/A | Boiling Point | N/A | |
| Molecular Formula | N/A | Melting Point | N/A | |
| MSDS | USA | Flash Point | N/A | |
Use of GLYCOL CHITOSANGlycol chitosan is a chitosan derivative with hydrophilic ethylene glycol branches. Glycol chitosan enhances membrane permeability and leadkage in Glycine max Harosoy 63W cells. Glycol chitosan is water-soluble, biocompatible and biodegradable[1][2][3]. Glycol chitosan inhibits E. coli, S. aureus and S. enteritidis growths with MIC values of 4 μg/mL, 32 μg/mL and <0.5 μg/mL, respectively[4]. |
| Name | GLYCOL CHITOSAN |
|---|---|
| Synonym | More Synonyms |
| Description | Glycol chitosan is a chitosan derivative with hydrophilic ethylene glycol branches. Glycol chitosan enhances membrane permeability and leadkage in Glycine max Harosoy 63W cells. Glycol chitosan is water-soluble, biocompatible and biodegradable[1][2][3]. Glycol chitosan inhibits E. coli, S. aureus and S. enteritidis growths with MIC values of 4 μg/mL, 32 μg/mL and <0.5 μg/mL, respectively[4]. |
|---|---|
| Related Catalog | |
| Target |
MIC: 4 μg/mL (E. coli), 32 μg/mL (S. aureus) and <0.5 μg/mL (S. enteritidis)[4] |
| In Vitro | Glycol chitosan derivatives have been successfully applied to deliver antimicrobial agents and anticancer drugs such as chemodrugs, genes, and photosensitizers (PSs), either by physical encapsulation or chemical conjugation. Glycol chitosan can be directly linked with hydrophobic drugs to generate amphiphilic compounds that can also form nanoparticles (NPs) for cell imaging and drug delivery. The use of Glycol chitosan derivatives for cell imaging and drug delivery has several advantages, including superb tumor-homing ability in the case of Glycol chitosan NPs based on enhanced permeability and retention (EPR) effect, low cytotoxicity, ease of chemical modification, great biocompatibility, and biodegradability[1]. The hydrophobic modification of Glycol chitosan is already confirmed, such as Glycol chitosan bearing a 5β-cholanic acid moiety and deoxycholic acid-Glycol chitosan, could self-assemble into nanoparticles, acting as a promising vehicle for hydrophobic drugs and genes[2]. |
| References |
| Appearance of Characters | crystalline |
|---|---|
| Storage condition | −20°C |
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| Hazard Codes | Xi |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
|
Effect of Chitosan on Membrane Permeability of Suspension-Cultured Glycine max and Phaseolus vulgaris Cells.
Plant Physiol. 70 , 1449-1454, (1982) Treatment of suspension-cultured Glycine max cv Harosoy 63 cells with soluble chitosan (20-500 micrograms per milliliter) increased membrane permeability as shown by leakage of electrolytes, protein, ... |
|
|
Pilot in vivo toxicological investigation of boron nitride nanotubes.
Int. J. Nanomedicine 7 , 19-24, (2012) Boron nitride nanotubes (BNNTs) have attracted huge attention in many different research fields thanks to their outstanding chemical and physical properties. During recent years, our group has pioneer... |
|
|
Barium Titanate Nanoparticles: Highly Cytocompatible Dispersions in Glycol-chitosan and Doxorubicin Complexes for Cancer Therapy.
Nanoscale Res. Lett. 5 , 1093-101, (2011) In the latest years, innovative nanomaterials have attracted a dramatic and exponentially increasing interest, in particular for their potential applications in the biomedical field. In this paper, we... |
| MFCD00131218 |

