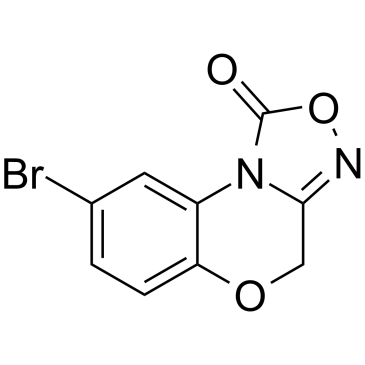
204326-43-2
| Name | 4h-8-bromo-1,2,4-oxadiazolo(3,4-d)benz(b)(1,4)oxazin-1-one |
|---|---|
| Synonyms |
4H-8-BROMO-1,2,4-OXADIAZOLO(3,4-D)BENZO(B)(1,4)OXAZIN-1-ONE
8-BROMO-4H-2,5-DIOXA-3,9B-DIAZA CYCLOPENTA[A]NAPHTHALEN-1-ONE Kartogenin 4H-8-Bromo-1,2,4-oxadiazolo[3,4-d]benz[b][1,4]oxazin-1-one NS-2028 8-Bromo-4H-[1,2,4]oxadiazolo[3,4-c][1,4]benzoxazin-1-one |
| Description | NS-2028 is a highly selective soluble Guanylyl Cyclase (sGC) inhibitor with IC50 values of 30 nM and 200 nM for basal and NO-stimulated enzyme activity[1]. NS-2028 inhibits soluble Guanylyl Cyclase activity in homogenates of mouse cerebellum and neuronal NO synthase with IC50 values of 17 nM and 20 nM[1]. NS-2028 inhibits 3-morpholino-sydnonimine (SIN-1)-elicited formation of cyclic GMP in human cultured umbilical vein endothelial cells with an IC50 of 30 nM[1]. NS-2028 is commonly used in the research of nitric oxide signaling pathways, it inhibits NO-dependent relaxant responses in non-vascular smooth muscle completely (1 μM)[1]. NS-2028 reduces vascular endothelial growth factor-induced angiogenesis and permeability[2]. |
|---|---|
| Related Catalog | |
| Target |
IC50: 30 nM (soluble Guanylyl Cyclase sGC)[1] |
| In Vitro | NS-2028 (10 μM; 24 hours) inhibits 25% cell number in comparation with those grown in the presence of vehicle[2]. NS-2028 (10 μM; 30 mins) attenuates VEGF-induced EC migration by inhibiting p38 MAPK activation[2]. Cell Proliferation Assay[2] Cell Line: HUVEC cells Concentration: 10 μM Incubation Time: 24 hours Result: Decreased cell numbers in culture. Western Blot Analysis[2] Cell Line: HUVEC cells Concentration: 10 μM Incubation Time: 30 mins Result: Attenuated VEGF-enhanced p38 phosphorylation. |
| In Vivo | NS-2028 (Deliver orally; 1 g/L; 8 days) exhibits a significant reduction of new vessel formation in the avascular rabbit cornea in response to VEGF pellet implants[2]. Animal Model: Rabbit[2] Dosage: 1 g/L Administration: Deliver orally; 1g/L; 8 days Result: Inhibits VEGF-induced angiogenesis in vivo. |
| References |
| Density | 2.1±0.1 g/cm3 |
|---|---|
| Boiling Point | 353.8±52.0 °C at 760 mmHg |
| Melting Point | 161-162ºC |
| Molecular Formula | C9H5BrN2O3 |
| Molecular Weight | 269.052 |
| Flash Point | 167.8±30.7 °C |
| Exact Mass | 267.948334 |
| PSA | 57.26000 |
| LogP | 0.27 |
| Vapour Pressure | 0.0±0.8 mmHg at 25°C |
| Index of Refraction | 1.778 |
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| Hazard Codes | Xi |
| RIDADR | NONH for all modes of transport |