77-19-0
| Name | dicyclomine |
|---|---|
| Synonyms |
Dicyclohexylhydroxyphenylstannane
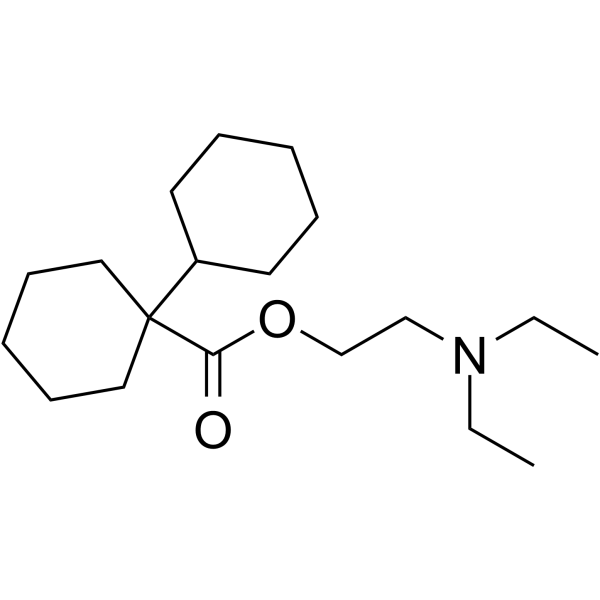
[1,1'-bicyclohexyl]-1-carboxylic acid 2-(diethylamino)ethyl ester Stannane,dicyclohexylhydroxyphenyl dicycloverine Dicyclohexylphenyltin hydroxide Bicyclohexyl-1-carbonsaeure-(2-diaethylamino-aethylester) bicyclohexyl-1-carboxylic acid-(2-diethylamino-ethyl ester) Dicyclomine |
| Description | Dicyclomine (Dicycloverine) is a potent and orally active muscarinic cholinergic receptors antagonist. Dicyclomine (Dicycloverine) shows high affinity for muscarinic M1 receptor subtype (Ki=5.1 nM) and M2 receptor subtype (Ki=54.6 nM) in brush-border membrane and basal plasma membranes, respectively[1]. Dicyclomine is an antispasmodic agent and relieves smooth muscle spasm of the gastrointestinal tract in vivo[2]. |
|---|---|
| Related Catalog | |
| In Vivo | Dicyclomine (Dicycloverine) (intraperitoneal injection; 8 mg/kg; daily) exacerbates the cognitive impairments in all the measurements. In addition, the memory impairments are worse in dicyclomine-treated 3xTg-AD mice compared to dicyclomine-treated NonTg mice[2]. Dicyclomine (Dicycloverine) (intraperitoneal injection; 2.0, 4.0, and 8.0 mg/kg; 7 days) produces a highly significant effect on performance in the paired-associates learning (PAL) task in mice.And systemic treatment at lower doses show behavioral impairments in mice in spatial tasks[3]. Animal Model: C57Bl/6 mice[1] Dosage: 2.0, 4.0, and 8.0 mg/kg Administration: Intraperitoneal injection; daily; 7 days Result: Produced impairments due to actions of the agent outside of the hippocampus. |
| References |
| Molecular Formula | C19H35NO2 |
|---|---|
| Molecular Weight | 309.48700 |
| Exact Mass | 309.26700 |
| PSA | 29.54000 |
| LogP | 4.40220 |
|
~% 
77-19-0 |
| Literature: Tilford; Van Campen; Shelton Journal of the American Chemical Society, 1947 , vol. 69, p. 2902,2904 |
|
~% 
77-19-0 |
| Literature: Yoshida; Iwashige Pharmaceutical Bulletin, 1955 , vol. 3, p. 417,420 |
|
~% 
77-19-0 |
| Literature: Verrier Bulletin de la Societe Chimique de France, 1959 , p. 1863 |
|
~% 
77-19-0 |
| Literature: Kopp; Tchoubar Bulletin de la Societe Chimique de France, 1952 , p. 84 |
|
~% 
77-19-0 |
| Literature: Kopp; Tchoubar Bulletin de la Societe Chimique de France, 1952 , p. 84 |
| Precursor 6 | |
|---|---|
| DownStream 0 | |