71376-34-6
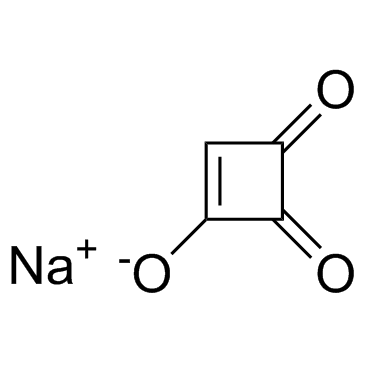
| Name | sodium,3,4-dioxocyclobuten-1-olate |
|---|---|
| Synonyms |
3-CYCLOBUTENE-1,2-DIONE,3-HYDROXY-,SODIUM SALT
1-Hydroxycyclobut-1-ene-3,4-dione Moniliformin sodium salt from Fusarium proliferatum 3-Hydroxy-3-cyclobutenedione sodium salt Moniliformin sodium salt 1-Hydroxycyclobut-1-ene-3,4-dione sodium salt Moniliformin (sodium salt) |
| Description | Moniliformin sodium salt is a potent, water-soluble mycotoxin isolate from Fusarium moniliforme. |
|---|---|
| Related Catalog | |
| In Vitro | Fusarium moniliforme NRRL 6322 produces about 600 mg of recoverable moniliformin, a mycotoxic metabolite, per kg of corn grit medium. Several strains of Fusarium moniliforme produce in laboratory culture more than 800 mg of moniliformin per kg of growth substrate[1]. Monocytes-derived macrophages exposed to moniliformin during the differentiation process present a decrease of endocytosis ability, and a decrease of CD71 and HLA-DR expression[2]. |
| In Vivo | Moniliformin is less toxic to mice, with an LD50 of 20.9 mg per kg for the females and 29.1 mg per kg of body weight for the males. As in the case of the chicks, mice surviving the toxin demonstrated no ill effects; the mice or chicks that died became recumbent in 4 to 6 h after treatment and died within 24 h. In 4-day-old chicken embryos, a sharp LD50 of 2.8 μg per embryo is obtained with no overt gross teratogenic effects in the survivors[1]. Rats treated with the highest dose of moniliformin show decreased activity followed by acute heart failure and death. The rats of the lower doses (<9mg/kg b.w.) show no signs of toxicity. The daily intake of moniliformin strongly reduces the phagocytic activity of neutrophils in all dose groups. The decrease continued in the satellite group during the follow-up period, indicating a severe impact on the immune system and a LOAEL value of 3mg/kg b.w. for moniliformin. Moniliformin is rapidly excreted into urine, ranging between 20.2 and 31.5% daily and shows no signs of accumulation. The concentration of moniliformin in faeces is less than 2%, which suggests efficient absorption from the gastrointestinal tract[3]. |
| Animal Admin | Rats: Moniliformin is prepared in water. In this experiment, 5 dose groups (3, 6, 9, 12 and 15 mg/kg moniliformin b.w.) of test animals are exposed to moniliformin for 28 days. Each group consists of 5 male Sprague-Dawley rats. The dose groups are determined based on our acute toxicity study of MON in rats. In addition, a control group administered with filtered tap water and two satellite groups (dosed 12 and 15 mg/kg b.w. moniliformin) are used. The two satellite groups are kept alive for an additional 14 days without treatment to detect possible delayed toxic effects and to follow up recovery[3]. Mice: Sterile aqueous solutions of moniliformin are injected intraperitoneally (0.2 mL) into five female and five male white mice, weighing about 25 g each, at concentrations equivalent to 0, 20, 25, 30, and 35 mg per kg of body weight. The mice are observed over a 4-day period, and LD50 values are determined[1]. |
| References |
| Boiling Point | 239.6ºC at 760 mmHg |
|---|---|
| Molecular Formula | C4HNaO3 |
| Molecular Weight | 120.03900 |
| Flash Point | 113ºC |
| Exact Mass | 119.98200 |
| PSA | 57.20000 |
| Storage condition | Store at 2-8 |
|
SECTION 1: Identification of the substance/mixture and of the company/undertaking Product identifiers Product name: Moniliformin sodium salt, from Fusarium proliferatum REACH No.: A registration number is not available for this substance as the substance or its uses are exempted from registration, the annual tonnage does not require a registration or the registration is envisaged for a later
registration deadline. CAS-No.: 71376-34-6 Relevant identified uses of the substance or mixture and uses advised against Identified uses: Laboratory chemicals, Manufacture of substances SECTION 2: Hazards identification Classification of the substance or mixture Classification according to Regulation (EC) No 1272/2008 Acute toxicity, Oral (Category 3), H301 For the full text of the H-Statements mentioned in this Section, see Section 16. Classification according to EU Directives 67/548/EEC or 1999/45/EC T ToxicR25 For the full text of the R-phrases mentioned in this Section, see Section 16. Label elements Labelling according Regulation (EC) No 1272/2008 Pictogram Signal wordDanger Hazard statement(s) H301Toxic if swallowed. Precautionary statement(s) P301 + P310IF SWALLOWED: Immediately call a POISON CENTER or doctor/ physician. Supplemental Hazardnone Statements Other hazards - none SECTION 3: Composition/information on ingredients Substances Chemical characterization : Natural product Synonyms: 1-Hydroxycyclobut-1-ene-3,4-dione Formula: C4HNaO3 Molecular Weight: 120,04 g/mol CAS-No.: 71376-34-6 Hazardous ingredients according to Regulation (EC) No 1272/2008 ComponentClassificationConcentration Moniliformin CAS-No.71376-34-6Acute Tox. 3; H301<= 100 % Hazardous ingredients according to Directive 1999/45/EC ComponentClassificationConcentration Moniliformin CAS-No.71376-34-6T, R25<= 100 % For the full text of the H-Statements and R-Phrases mentioned in this Section, see Section 16 SECTION 4: First aid measures Description of first aid measures General advice Consult a physician. Show this safety data sheet to the doctor in attendance. If inhaled If breathed in, move person into fresh air. If not breathing, give artificial respiration. Consult a physician. In case of skin contact Wash off with soap and plenty of water. Take victim immediately to hospital. Consult a physician. In case of eye contact Flush eyes with water as a precaution. If swallowed Never give anything by mouth to an unconscious person. Rinse mouth with water. Consult a physician. Most important symptoms and effects, both acute and delayed The most important known symptoms and effects are described in the labelling (see section 2.2) and/or in section 11 Indication of any immediate medical attention and special treatment needed no data available SECTION 5: Firefighting measures Extinguishing media Suitable extinguishing media Use water spray, alcohol-resistant foam, dry chemical or carbon dioxide. Special hazards arising from the substance or mixture Carbon oxides, Sodium oxides Advice for firefighters Wear self contained breathing apparatus for fire fighting if necessary. Further information no data available SECTION 6: Accidental release measures Personal precautions, protective equipment and emergency procedures Wear respiratory protection. Avoid dust formation. Avoid breathing vapours, mist or gas. Ensure adequate ventilation. Evacuate personnel to safe areas. Avoid breathing dust. For personal protection see section 8. Environmental precautions Prevent further leakage or spillage if safe to do so. Do not let product enter drains. Methods and materials for containment and cleaning up Pick up and arrange disposal without creating dust. Sweep up and shovel. Keep in suitable, closed containers for disposal. Reference to other sections For disposal see section 13. SECTION 7: Handling and storage Precautions for safe handling Avoid contact with skin and eyes. Avoid formation of dust and aerosols. Provide appropriate exhaust ventilation at places where dust is formed. For precautions see section 2.2. Conditions for safe storage, including any incompatibilities Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Recommended storage temperature: 2 - 8 °C Specific end use(s) A part from the uses mentioned in section 1.2 no other specific uses are stipulated SECTION 8: Exposure controls/personal protection Control parameters Components with workplace control parameters Exposure controls Appropriate engineering controls Avoid contact with skin, eyes and clothing. Wash hands before breaks and immediately after handling the product. Personal protective equipment Eye/face protection Face shield and safety glasses Use equipment for eye protection tested and approved under appropriate government standards such as NIOSH (US) or EN 166(EU). Skin protection Handle with gloves. Gloves must be inspected prior to use. Use proper glove removal technique (without touching glove's outer surface) to avoid skin contact with this product. Dispose of contaminated gloves after use in accordance with applicable laws and good laboratory practices. Wash and dry hands. The selected protective gloves have to satisfy the specifications of EU Directive 89/686/EEC and the standard EN 374 derived from it. Full contact Material: Nitrile rubber Minimum layer thickness: 0,11 mm Break through time: 480 min Material tested:Dermatril® (KCL 740 / Z677272, Size M) Splash contact Material: Nitrile rubber Minimum layer thickness: 0,11 mm Break through time: 480 min Material tested:Dermatril® (KCL 740 / Z677272, Size M) data source: KCL GmbH, D-36124 Eichenzell, phone +49 (0)6659 87300, test method: EN374 If used in solution, or mixed with other substances, and under conditions which differ from EN 374, contact the supplier of the CE approved gloves. This recommendation is advisory only and must be evaluated by an industrial hygienist and safety officer familiar with the specific situation of anticipated use by our customers. It should not be construed as offering an approval for any specific use scenario. Body Protection Complete suit protecting against chemicals, The type of protective equipment must be selected according to the concentration and amount of the dangerous substance at the specific workplace. Respiratory protection Where risk assessment shows air-purifying respirators are appropriate use a full-face particle respirator type N99 (US) or type P2 (EN 143) respirator cartridges as a backup to engineering controls. If the respirator is the sole means of protection, use a full-face supplied air respirator. Use respirators and components tested and approved under appropriate government standards such as NIOSH (US) or CEN (EU). Control of environmental exposure Prevent further leakage or spillage if safe to do so. Do not let product enter drains. SECTION 9: Physical and chemical properties Information on basic physical and chemical properties a) AppearanceForm: solid b) Odourno data available c) Odour Thresholdno data available d) pHno data available e) Melting point/freezingno data available point f) Initial boiling point and no data available boiling range g) Flash pointno data available h) Evapouration rateno data available i) Flammability (solid, gas) no data available j) Upper/lowerno data available flammability or explosive limits k) Vapour pressureno data available l) Vapour densityno data available m) Relative densityno data available n) Water solubilityno data available o) Partition coefficient: n- no data available octanol/water p) Auto-ignitionno data available temperature q) Decompositionno data available temperature r) Viscosityno data available s) Explosive propertiesno data available t) Oxidizing propertiesno data available Other safety information no data available SECTION 10: Stability and reactivity Reactivity no data available Chemical stability Stable under recommended storage conditions. Possibility of hazardous reactions no data available Conditions to avoid no data available Incompatible materials Strong oxidizing agents Hazardous decomposition products Other decomposition products - no data available In the event of fire: see section 5 SECTION 11: Toxicological information Information on toxicological effects Acute toxicity LD50 Intraperitoneal - mouse - 21 mg/kg Remarks: Behavioral:Altered sleep time (including change in righting reflex). Skin corrosion/irritation no data available Serious eye damage/eye irritation no data available Respiratory or skin sensitisation no data available Germ cell mutagenicity no data available Carcinogenicity IARC:No component of this product present at levels greater than or equal to 0.1% is identified as probable, possible or confirmed human carcinogen by IARC. Reproductive toxicity no data available Specific target organ toxicity - single exposure no data available Specific target organ toxicity - repeated exposure no data available Aspiration hazard no data available Additional Information RTECS: GU1815000 To the best of our knowledge, the chemical, physical, and toxicological properties have not been thoroughly investigated. SECTION 12: Ecological information Toxicity no data available Persistence and degradability no data available Bioaccumulative potential no data available Mobility in soil no data available Results of PBT and vPvB assessment PBT/vPvB assessment not available as chemical safety assessment not required/not conducted Other adverse effects no data available SECTION 13: Disposal considerations Waste treatment methods Product Offer surplus and non-recyclable solutions to a licensed disposal company. Dissolve or mix the material with a combustible solvent and burn in a chemical incinerator equipped with an afterburner and scrubber. Contaminated packaging Dispose of as unused product. SECTION 14: Transport information UN number ADR/RID: 2811IMDG: 2811IATA: 2811 UN proper shipping name ADR/RID: TOXIC SOLID, ORGANIC, N.O.S. (Moniliformin) IMDG: TOXIC SOLID, ORGANIC, N.O.S. (Moniliformin) IATA:Toxic solid, organic, n.o.s. (Moniliformin) Transport hazard class(es) ADR/RID: 6.1IMDG: 6.1IATA: 6.1 Packaging group ADR/RID: IIIMDG: IIIATA: II Environmental hazards ADR/RID: noIMDG Marine pollutant: noIATA: no Special precautions for user no data available SECTION 15 - REGULATORY INFORMATION N/A SECTION 16 - ADDITIONAL INFORMATION N/A |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Symbol |

GHS06 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H301 |
| Precautionary Statements | P301 + P310 |
| Personal Protective Equipment | Eyeshields;Faceshields;Gloves;type P2 (EN 143) respirator cartridges |
| Hazard Codes | T: Toxic; |
| Risk Phrases | 25 |
| Safety Phrases | 45 |
| RIDADR | UN 2811 6.1/PG 2 |
| RTECS | GU1815000 |