23094-69-1
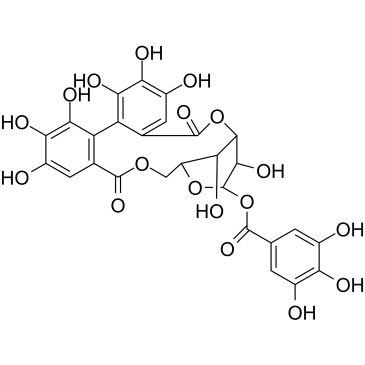
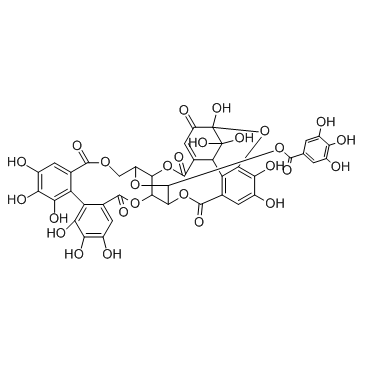
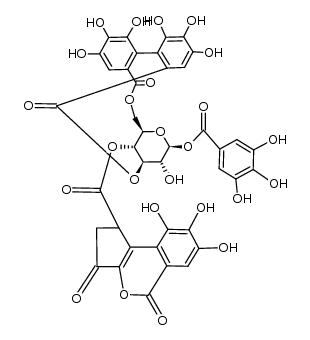
| Name | corilagin |
|---|---|
| Synonyms |
Benzoic acid, 3,4,5-trihydroxy-, (8R,10S,11R,12S,19R)-5,7,8,11,12,14-hexahydro-1,2,3,11,16,17,18,19-octahydroxy-5,14-dioxo-8,12-methano-10H-dibenzo[j,l][1,4,8]trioxacyclotetradecin-10-yl ester
(1S,19R,21S,22R,23R)-6,7,8,11,12,13,22,23-Octahydroxy-3,16-dioxo-2,17,20-trioxatetracyclo[17.3.1.0.0]tricosa-4,6,8,10,12,14-hexaen-21-yl 3,4,5-trihydroxybenzoate corilagin 1-O-Galloyl-3,6-hexahydroxydiphenol-b-D-Glucopyranose 1-O-Galloyl-3,6-hexahydroxydiphenol--D-Glucopyranose |
| Description | Corilagin, a gallotannin, is isolated from Caesalpinia coriaria (Jacq.) Willd. Corilagin inhibits activity of reverse transcriptase of RNA tumor viruses. Corilagin inhibits the growth of Staphylococcus aureus with a MIC of 25 μg/mL. Corilagin shows good anti-tumor activity on hepatocellular carcinoma and ovarian cancer. Corilagin shows a low level of toxicity toward normal cells and tissues[1]. |
|---|---|
| Related Catalog | |
| References |
[1]. Li X, et al. Corilagin, a promising medicinal herbal agent. Biomed Pharmacother. 2018 Mar;99:43-50. |
| Density | 2.1±0.1 g/cm3 |
|---|---|
| Boiling Point | 1280.8±65.0 °C at 760 mmHg |
| Melting Point | >200ºC dec |
| Molecular Formula | C27H22O18 |
| Molecular Weight | 634.453 |
| Flash Point | 418.8±27.8 °C |
| Exact Mass | 634.080627 |
| PSA | 310.66000 |
| LogP | 2.34 |
| Vapour Pressure | 0.0±0.3 mmHg at 25°C |
| Index of Refraction | 1.881 |
| Storage condition | -20°C Freezer |
| Water Solubility | H2O: soluble1mg/mL, clear, colorless |
| RIDADR | NONH for all modes of transport |
|---|
| Precursor 6 | |
|---|---|
| DownStream 1 | |