414910-30-8
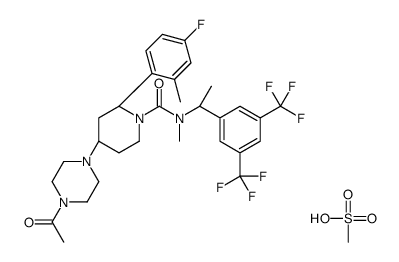
| Name | (2R,4S)-4-(4-acetylpiperazin-1-yl)-N-[(1R)-1-[3,5-bis(trifluoromethyl)phenyl]ethyl]-2-(4-fluoro-2-methylphenyl)-N-methylpiperidine-1-carboxamide,methanesulfonic acid |
|---|---|
| Synonyms |
Rezonic
UNII-7VSV9BL497 (2R,4S)-4-(4-ACETYL-(PIPERAZIN-1-YL))-N-{(1R)-1-[3,5-BIS(TRIFLUORO-METHYL)PHENYL]-ETHYL}-2-(4-FLUORO-2-METHYLPHENYL)-N-METHYL-1-PIPERIDINE-CARBOXAMIDE MONOMETHANE-SULFONATE Zunrisa Casopitant mesylate Casopitant mesylate (USAN) |
| Description | Casopitant mesylate (GW679769B) is a potent, selective, brain permeable and orally active neurokinin 1 (NK1) receptor antagonist. Casopitant mesylate is a second in the class of antiemetics that acts to antagonise the emetogenic effect of substance P. Casopitant mesylate is also a substrate and a weak-to-moderate inhibitor of CYP3A4. Casopitant mesylate can be used for chemotherapy-induced nausea and vomiting (CINV) and postoperative nausea and vomiting (PONV)[1][2]. |
|---|---|
| Related Catalog | |
| Target |
NK1 CYP3A4 |
| In Vivo | In a ferret-model of Cisplatin- induced emesis, Casopitant (GW679769) inhibits retching and vomiting and reduced nausea-like behaviours in a dose-dependent manner. The pharmacokinetics and brain penetration of casopitant are studied in the ferret-model of cisplatin-induced emesis. Following a single intraperitoneal dose, radioactive labeled Casopitant ([14C]Casopitant) is rapidly absorbed, with plasma and brain concentrations being approximately equal at two hours post-dosing. [14C]Casopitant is found in the brain as the parent compound and two major oxidative metabolites (M1 and M2), accounting for approximately 76%, 19%, and 3% of the radioactivity, respectively; suggesting that the pharmacologic activity of Casopitant in the ferret is largely attributable to the parent compound. Casopitant possesses a high affinity for brain NK1 receptors in the ferret[2]. |
| References |
| Molecular Formula | C31H39F7N4O5S |
|---|---|
| Molecular Weight | 712.71900 |
| Exact Mass | 712.25300 |
| PSA | 109.85000 |
| LogP | 7.05280 |