88373-72-2
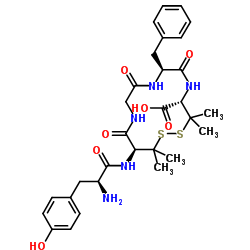
| Name | (4R,7S,13S)-13-[[(2S)-2-amino-3-(4-hydroxyphenyl)propanoyl]amino]-7-benzyl-3,3,14,14-tetramethyl-6,9,12-trioxo-1,2-dithia-5,8,11-triazacyclotetradecane-4-carboxylic acid |
|---|---|
| Synonyms |
[D-Pen2,Pen5]-Enkephalin
(4S,7S,13S)-7-Benzyl-3,3,14,14-tetramethyl-6,9,12-trioxo-13-(L-tyrosylamino)-1,2-dithia-5,8,11-triazacyclotetradecane-4-carboxylic acid DPDPE |
| Description | [DPen2, Pen5] Enkephalin is a δ-opioid receptor selective analog of [Leu5]-Enkephalin (HY-P0288)[1]. |
|---|---|
| Related Catalog | |
| Target |
δ Opioid Receptor/DOR |
| In Vivo | [DPen2, Pen5] Enkephalin (0-3.32 μg/kg, IP) impairs acquisition of an automated jump-up avoidance response in rats and acquisition of a one-way active avoidance response in mice[1]. Animal Model: Male Sprague-Dawley rats (260-280 g)[1] Dosage: 0, 1.16, 11.6 μg/kg Administration: IP, administered on day 2 prior to presentation of eight training trials, on subsequent one-way avoidance responding. Result: Produced a significant impairment of avoidance performance at 1.16 ug/kg, while the 11.6 ug/kg dose was without significant effect. Animal Model: Male Sprague-Dawley rats (260-280 g)[1] Dosage: 0, 0.332, 3.32 μg/kg Administration: IP, administered on day 1 immediately after presentation of two escape-only trials, on subsequent day 2 one-way avoidance responding in the second experiment Result: Produced a significant enhancement of avoidance performance at 0.332 ug/kg, while the 3.32 ug/kg dose was without significant effect. |
| Density | 1.4±0.1 g/cm3 |
|---|---|
| Boiling Point | 1038.6±65.0 °C at 760 mmHg |
| Molecular Formula | C30H39N5O7S2 |
| Molecular Weight | 645.790 |
| Flash Point | 581.9±34.3 °C |
| Exact Mass | 645.229065 |
| PSA | 250.55000 |
| LogP | 2.24 |
| Vapour Pressure | 0.0±0.3 mmHg at 25°C |
| Index of Refraction | 1.653 |
| WGK Germany | 3 |
|---|