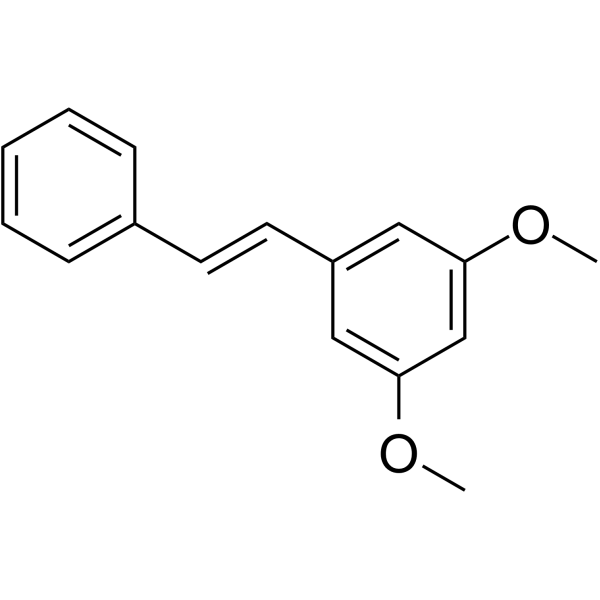
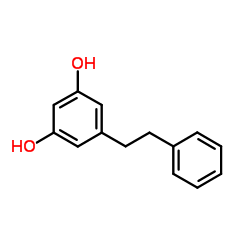
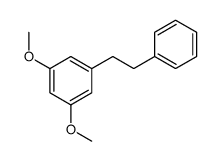
35302-70-6
| Name | pinosylvin mono methyl ether |
|---|---|
| Synonyms |
(E)-3-Hydroxy-5-methoxystilbene,(E)-3-Methoxy-5-(2-phenylethenyl)phenol,5-Methoxy-3-stilbenol
3-Methoxy-5-[(E)-2-phenylvinyl]phenol O-Methyl-pinosylvin pinosylvin monomethyl ether 3-methoxy-5-[(E)-2-phenylethenyl]phenol pinosylvin-O-methyl ether 5-Hydroxy-3-methoxy-trans-stilben 5-Methoxy-3-stilbenol 3-hydroxy-5-methoxystilbene |
| Description | Pinosylvin monomethyl ether has antibacterial effect[1] and fungicidal activity[2]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 405.8±24.0 °C at 760 mmHg |
| Molecular Formula | C15H14O2 |
| Molecular Weight | 226.27 |
| Flash Point | 245.6±7.8 °C |
| Exact Mass | 226.099380 |
| PSA | 29.46000 |
| LogP | 4.34 |
| Vapour Pressure | 0.0±1.0 mmHg at 25°C |
| Index of Refraction | 1.662 |
| Hazard Codes | Xn,N |
|---|---|
| Risk Phrases | 22-36-51/53 |
| Safety Phrases | 26-61 |
| RIDADR | UN 3077 9 / PGIII |
|
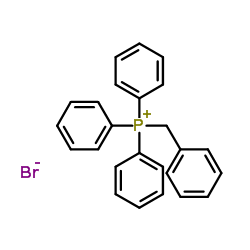
~73% 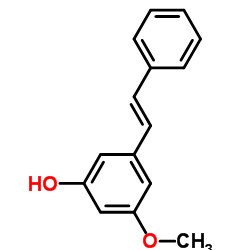
35302-70-6 |
| Literature: Kabir, M. Shahjahan; Engelbrecht, Kathleen; Polanowski, Rebecca; Krueger, Sarah M.; Ignasiak, Rachel; Rott, Marc; Schwan, William R.; Stemper, Mary E.; Reed, Kurt D.; Sherman, David; Cook, James M.; Monte, Aaron Bioorganic and Medicinal Chemistry Letters, 2008 , vol. 18, # 21 p. 5745 - 5749 |
|
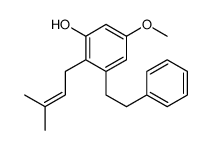
~44% 
35302-70-6 |
| Literature: Chemical and Pharmaceutical Bulletin, , vol. 40, # 5 p. 1130 - 1136 |
|
~% 
35302-70-6 |
| Literature: Bioorganic and Medicinal Chemistry Letters, , vol. 14, # 23 p. 5895 - 5898 |
|
~% 
35302-70-6 |
| Literature: Bioorganic and Medicinal Chemistry Letters, , vol. 14, # 23 p. 5895 - 5898 |
|
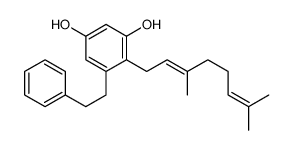
~% 
35302-70-6 |
| Literature: Bioorganic and Medicinal Chemistry Letters, , vol. 14, # 23 p. 5895 - 5898 |
|
~% 
35302-70-6 |
| Literature: Chemical and Pharmaceutical Bulletin, , vol. 40, # 5 p. 1130 - 1136 |
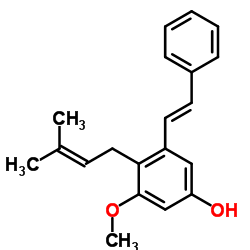
| Precursor 5 | |
|---|---|
| DownStream 7 | |