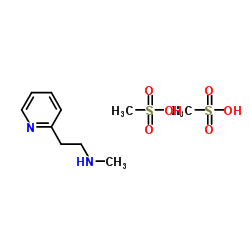
54856-23-4
| Name | Betahistine Mesylate |
|---|---|
| Synonyms |
2-Pyridineethanamine, N-methyl-, methanesulfonate (1:2)
N-Methyl-2-pyridineethanamine Dimethanesulfonate MFCD00211321 N-Methyl-2-(2-pyridinyl)ethanamine methanesulfonate (1:2) EINECS 259-377-7 N-Methyl-2-(pyridin-2-yl)ethanamine methanesulfonate (1:2) methanesulfonic acid,N-methyl-2-pyridin-2-ylethanamine |
| Description | Betahistine mesylate is an orally active histamine H1 receptor agonist and a H3 receptor antagonist[1]. Betahistine mesylate is used for the study of rheumatoid arthritis (RA)[3]. |
|---|---|
| Related Catalog | |
| In Vitro | Betahistine mesylate (0-10 μM) inhibits [125I]iodoproxyfan binding to membranes of CHO (rH3(445)R) and CHO (hH3(445)R) cells with IC50 values of 1.9 μM and 3.3 μM, respectively. Lead to Ki values of 1.4 μM and 2.5 μM, respectively[2]. Betahistine mesylate (0-10 μM) has a regulating function on cAMP formation in CHO (rH3(445)R), CHO (rH3(413)R), and CHO (hH3(445)R) cells. At low concentrations, Betahistine mesylate behaves an apparent inverse agonist, and progressively enhances cAMP formation with EC50 values of 0.1 nM, 0.05 nM and 0.3 nM, respectively. In contrast, at concentrations higher than 10 nM, Betahistine mesylate inhibits cAMP formation with an EC50 value of 0.1 μM in CHO (rH3(445)R) and full agonist activity[2]. |
| In Vivo | Betahistine mesylate (intraperitoneal or oral administration; 0.1-30 mg/kg; single dose) with acute administration has increased tele-methylhistamine (t-MeHA) levels with an ED50 of 0.4 mg/kg, indicating the inverse agonism. Besides, after acute oral administration, it increases t-MeHA levels with an ED50 of 2 mg/kg in male Swissmice[2]. Betahistine mesylate (oral adminstration; 1 and 5 mg/kg; daily for 3 weeks) attenuates the severity of arthritis and reduces the levels of pro-inflammatory cytokines in the paw tissues of CIA mice[3]. Animal Model: Collagen-induced arthritis (CIA) DBA/1 male mouse model[3] Dosage: 1 mg/kg; 5mg/kg Administration: Oral adminstration; day 21 to day 42 after a 21-day CIA induction Result: Ameliorated mouse CIA by decreasing joint destruction. |
| References |
| Boiling Point | 210.9ºC at 760 mmHg |
|---|---|
| Melting Point | 112°C |
| Molecular Formula | C10H20N2O6S2 |
| Molecular Weight | 328.406 |
| Flash Point | 96.7ºC |
| Exact Mass | 328.076263 |
| PSA | 150.42000 |
| LogP | 2.40400 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| RTECS | UT2970200 |
|---|---|
| HS Code | 2933399090 |
| HS Code | 2933399090 |
|---|---|
| Summary | 2933399090. other compounds containing an unfused pyridine ring (whether or not hydrogenated) in the structure. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |