TBPB

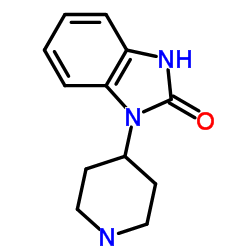
TBPB structure
|
Common Name | TBPB | ||
|---|---|---|---|---|
| CAS Number | 634616-95-8 | Molecular Weight | 404.548 | |
| Density | 1.2±0.1 g/cm3 | Boiling Point | N/A | |
| Molecular Formula | C25H32N4O | Melting Point | N/A | |
| MSDS | USA | Flash Point | N/A | |
Use of TBPBTBPB is an allosteric M1 mAChR agonist(EC50=289 nM) that regulates amyloid processing and produces antipsychotic-like activity in rats.IC50 value: 289 nM(EC50) [2]Target: M1 mAChR agonistin vitro: TBPB activates M(1) through an allosteric site rather than the orthosteric acetylcholine binding site, which is likely critical for its unprecedented selectivity. Whole-cell patch-clamp recordings demonstrated that activation of M(1) by TBPB potentiates NMDA receptor currents in hippocampal pyramidal cells but does not alter excitatory or inhibitory synaptic transmission, responses thought to be mediated by M(2) and M(4) [1]. in vivo: TBPB was efficacious in models predictive of antipsychotic-like activity in rats at doses that did not produce catalepsy or peripheral adverse effects of other mAChR agonists [1]. |
| Name | 1-[1'-(2-Methylbenzyl)-1,4'-bipiperidin-4-yl]-1,3-dihydro-2H-benz imidazol-2-one |
|---|---|
| Synonym | More Synonyms |
| Description | TBPB is an allosteric M1 mAChR agonist(EC50=289 nM) that regulates amyloid processing and produces antipsychotic-like activity in rats.IC50 value: 289 nM(EC50) [2]Target: M1 mAChR agonistin vitro: TBPB activates M(1) through an allosteric site rather than the orthosteric acetylcholine binding site, which is likely critical for its unprecedented selectivity. Whole-cell patch-clamp recordings demonstrated that activation of M(1) by TBPB potentiates NMDA receptor currents in hippocampal pyramidal cells but does not alter excitatory or inhibitory synaptic transmission, responses thought to be mediated by M(2) and M(4) [1]. in vivo: TBPB was efficacious in models predictive of antipsychotic-like activity in rats at doses that did not produce catalepsy or peripheral adverse effects of other mAChR agonists [1]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Molecular Formula | C25H32N4O |
| Molecular Weight | 404.548 |
| Exact Mass | 404.257599 |
| PSA | 44.53000 |
| LogP | 4.96 |
| Index of Refraction | 1.623 |
| InChIKey | CWPKTBMRVATCBL-UHFFFAOYSA-N |
| SMILES | Cc1ccccc1CN1CCC(N2CCC(n3c(=O)[nH]c4ccccc43)CC2)CC1 |
| Storage condition | 2-8℃ |
| RIDADR | NONH for all modes of transport |
|---|
|
~75% 
TBPB CAS#:634616-95-8 |
| Literature: Rasheed, Mohammed Abdul; Shaik, Nagul Meera; Nirogi, Ramakrishna Synthetic Communications, 2013 , vol. 43, # 13 p. 1796 - 1801 |
|
~82% 
TBPB CAS#:634616-95-8 |
| Literature: Bridges, Thomas M.; Brady, Ashley E.; Phillip Kennedy; Nathan Daniels; Miller, Nicole R.; Kim, Kwango; Breininger, Micah L.; Gentry, Patrick R.; Brogan, John T.; Jones, Carrie K.; Jeffrey Conn; Lindsley, Craig W. Bioorganic and Medicinal Chemistry Letters, 2008 , vol. 18, # 20 p. 5439 - 5442 |
|
~% 
TBPB CAS#:634616-95-8 |
| Literature: Rasheed, Mohammed Abdul; Shaik, Nagul Meera; Nirogi, Ramakrishna Synthetic Communications, 2013 , vol. 43, # 13 p. 1796 - 1801 |
|
~% 
TBPB CAS#:634616-95-8 |
| Literature: Rasheed, Mohammed Abdul; Shaik, Nagul Meera; Nirogi, Ramakrishna Synthetic Communications, 2013 , vol. 43, # 13 p. 1796 - 1801 |
|
~% 
TBPB CAS#:634616-95-8 |
| Literature: Rasheed, Mohammed Abdul; Shaik, Nagul Meera; Nirogi, Ramakrishna Synthetic Communications, 2013 , vol. 43, # 13 p. 1796 - 1801 |
|
Cotton-based diagnostic devices.
Sci. Rep. 4 , 6976, (2014) A good diagnostic procedure avoids wasting medical resources, is easy to use, resists contamination, and provides accurate information quickly to allow for rapid follow-up therapies. We developed a no... |
|
|
Current and novel therapeutic molecules and targets in Alzheimer's disease.
J. Formos. Med. Assoc. 115 , 10-Mar, (2016) Alzheimer's disease (AD) is a neurodegenerative disorder in which the death of brain cells causes memory loss and cognitive decline, i.e., dementia. The disease starts with mild symptoms and gradually... |
|
Name: Agonist activity at muscarinic M3 receptor
Source: ChEMBL
Target: Muscarinic acetylcholine receptor M3
External Id: CHEMBL983584
|
|
Name: Displacement of [3H]NMS from rat muscarinic M1 receptor expressed in CHO cells after ...
Source: ChEMBL
Target: Muscarinic acetylcholine receptor M1
External Id: CHEMBL2319076
|
|
Name: Agonist activity at muscarinic M2 receptor
Source: ChEMBL
Target: Muscarinic acetylcholine receptor M2
External Id: CHEMBL983583
|
|
Name: Agonist activity at muscarinic M5 receptor
Source: ChEMBL
Target: Muscarinic acetylcholine receptor M5
External Id: CHEMBL983586
|
|
Name: Primary qHTS assay for small molecule inhibitors of Inositol hexaphosphate kinase 1 (...
Source: NCGC
External Id: IP6K1-p1
|
|
Name: Agonist activity at muscarinic M4 receptor
Source: ChEMBL
Target: Muscarinic acetylcholine receptor M4
External Id: CHEMBL983585
|
|
Name: Agonist activity at muscarinic M1 receptor relative to carbachol
Source: ChEMBL
Target: Muscarinic acetylcholine receptor M1
External Id: CHEMBL983580
|
|
Name: Agonist activity at muscarinic M1 receptor
Source: ChEMBL
Target: Muscarinic acetylcholine receptor M1
External Id: CHEMBL983579
|
|
Name: Inhibition of dopamine D2 receptor
Source: ChEMBL
Target: D(2) dopamine receptor
External Id: CHEMBL983581
|
|
Name: Cytochrome P450 family 3 subfamily A member 4 (CYP3A4) small molecule antagonists: lu...
Source: NCGC
External Id: CYP3A4437
|
| 2-Pyrrolo-1,3,2-dioxa-phospholan |
| 1-[1'-(2-Methylbenzyl)-1,4'-bipiperidin-4-yl]-1,3-dihydro-2H-benzimidazol-2-one |
| 2-pyrrol-1-yl-1,3,2-dioxaphospholan |
| TBPB |