Bioorganic & Medicinal Chemistry Letters
2008-12-15
Rational design of novel glycomimetics: inhibitors of concanavalin A.
Karen T Welch, Trent A Turner, Callie E Preast
Index: Bioorg. Med. Chem. Lett. 18 , 6573-5, (2008)
Full Text: HTML
Abstract
A virtual screening approach was used to identify new glycomimetics. The National Cancer Institute Diversity Set was docked into the carbohydrate binding site of the lectin concanavalin A (ConA). The resulting poses were analyzed and 19 molecules were tested for inhibition with an enzyme-linked lectin assay (ELLA). Eight of the 19 molecules inhibited ConA-carbohydrate binding. The two most potent inhibitors have IC(50) values that are an order of magnitude smaller than the monosaccharide methyl alpha-D-mannopyranoside.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
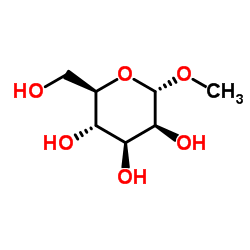 |
Methyl α-D-mannopyranoside
CAS:617-04-9 |
C7H14O6 |
Related Articles:
More...
|
Synthesis of glycopeptide dendrimers, dimerization and affin...
2011-05-01 [Bioorg. Med. Chem. 19 , 2879-87, (2011)] |
|
Redox-responsive and calcium-dependent switching of glycosyl...
2005-06-02 [Bioorg. Med. Chem. Lett. 15 , 2707-10, (2005)] |
|
BK Virus replication in vitro: limited effect of drugs inter...
2007-12-01 [Antimicrob. Agents Chemother. 51 , 4492-4, (2007)] |
|
Towards a stable noeuromycin analog with a D-manno configura...
2012-01-15 [Bioorg. Med. Chem. 20 , 641-9, (2012)] |
|
Prevention of colonization of the urinary tract of mice with...
1979-03-01 [J. Infect. Dis. 139 , 329-32, (1979)] |