| Structure | Name/CAS No. | Articles |
|---|---|---|
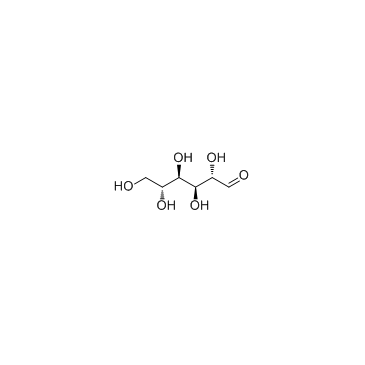 |
D-Mannose
CAS:3458-28-4 |
|
 |
4-Nitrophenyl α-D-mannopyranoside
CAS:10357-27-4 |
|
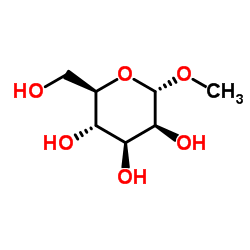 |
Methyl α-D-mannopyranoside
CAS:617-04-9 |