Journal of the American Chemical Society
2006-12-20
Axially chiral guanidine as highly active and enantioselective catalyst for electrophilic amination of unsymmetrically substituted 1,3-dicarbonyl compounds.
Masahiro Terada, Megumi Nakano, Hitoshi Ube
Index: J. Am. Chem. Soc. 128 , 16044, (2006)
Full Text: HTML
Abstract
A newly designed axially chiral guanidine is found to function as an effective platform for asymmetric induction at the alpha-carbon of unsymmetrically substituted 1,3-dicarbonyl compounds. Highly efficient and enantioselective electrophilic amination of various 1,3-dicarbonyl compounds with azodicarboxylate was successfully achieved using the present chiral guanidine catalyst, which provides efficient access to the construction of nitrogen-substituted quaternary stereocenters in an optically active form.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
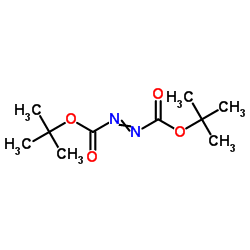 |
Di-tert-Butyl azodicarboxylate
CAS:870-50-8 |
C10H18N2O4 |
Related Articles:
More...
|
Chirally aminated 2-naphthols--organocatalytic synthesis of ...
2006-02-06 [Angew. Chem. Int. Ed. Engl. 45(7) , 1147-1151, (2006)] |
|
Catalytic Enantioselective Reaction of α-Aminoacetonitriles ...
2015-07-06 [Angew. Chem. Int. Ed. Engl. 54(28) , 8198-202, (2015)] |
|
Direct amination of α-substituted nitroacetates using di-ter...
2012-02-14 [Org. Biomol. Chem. 10(6) , 1158-61, (2012)] |
|
M. Kiankarimi et al.
[Tetrahedron Lett. 40 , 4497 , (1999)] |
|
J.L. Vicario et al.
[Tetrahedron Lett. 40 , 7123 , (1999)] |