Secondary metabolites from the roots of Neolitsea daibuensis and their anti-inflammatory activity.
Su-Ling Wong, Hsun-Shuo Chang, Guei-Jane Wang, Michael Y Chiang, Hung-Yi Huang, Chu-Huang Chen, Shiow-Chwen Tsai, Chu-Hung Lin, Ih-Sheng Chen
Index: J. Nat. Prod. 74(12) , 2489-96, (2011)
Full Text: HTML
Abstract
Bioassay-guided fractionation of the roots of Neolitsea daibuensis afforded three new β-carboline alkaloids, daibucarbolines A-C (1-3), three new sesquiterpenoids, daibulactones A and B (4 and 5) and daibuoxide (6), and 20 known compounds. The structures of 1-6 were determined by spectroscopic analysis and single-crystal X-ray diffraction. Daibucarboline A (1), isolinderalactone (7), 7-O-methylnaringenin (8), and prunetin (9) exhibited moderate iNOS inhibitory activity, with IC₅₀ values of 18.41, 0.30, 19.55, and 10.50 μM, respectively.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
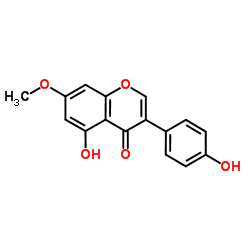 |
Prunetin
CAS:552-59-0 |
C16H12O5 |
|
Preparation and evaluation of surface-bonded tricationic ion...
2015-05-29 [J. Chromatogr. A. 1396 , 62-71, (2015)] |
|
Absorption of red clover isoflavones in human subjects: resu...
2010-06-01 [Br. J. Nutr. 103(11) , 1569-72, (2010)] |
|
The phytoestrogen prunetin affects body composition and impr...
2016-02-01 [FASEB J. 30 , 948-58, (2016)] |
|
Dalsympathetin--a new isoflavone gentiobioside from Dalbergi...
2006-02-01 [Nat. Prod. Res. 20(2) , 195-200, (2006)] |
|
Effects of prunetin on the proteolytic activity, secretion a...
2016-03-01 [Korean J. Physiol. Pharmacol. 20 , 221-8, (2016)] |