| Structure | Name/CAS No. | Articles |
|---|---|---|
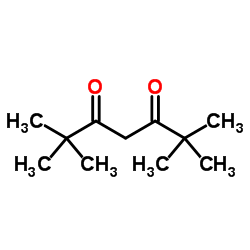 |
2,2,6,6-Tetramethylheptane-3,5-dione
CAS:1118-71-4 |
Elizabeth Buck, Zhiguo Jake Song, David Tschaen, Peter G Dormer, R P Volante, Paul J Reider
Index: Org. Lett. 4(9) , 1623-6, (2002)
Full Text: HTML
[reaction: see text]. In the copper salt catalyzed ether formation from aryl bromides or iodides and phenols, 2,2,6,6-tetramethylheptane-3,5-dione (TMHD) was found to greatly accelerate the ordinarily difficult reaction, making it occur under more moderate temperatures and reaction times. A series of aryl halides and phenols were shown to form ethers in NMP as the solvent, cesium carbonate as the base, and CuCl and TMHD as the catalysts. The reaction was shown to tolerate electron-rich aryl bromides and electron-neutral phenols.
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
 |
2,2,6,6-Tetramethylheptane-3,5-dione
CAS:1118-71-4 |
C11H20O2 |
|
26Mg as a probe in research on the role of magnesium in nutr...
1982-10-01 [Fed. Proc. 41(10) , 2709-13, (1982)] |
|
Ultrafine 239PuO2 aerosol generation, characterization and s...
1980-09-01 [Health Phys. 39(3) , 505-19, (1980)] |
|
Wavelength dependent photofragmentation patterns of tris(2,2...
2006-06-29 [J. Phys. Chem. A 110(25) , 7751-4, (2006)] |
|
[Tetrahedron 48 , 6909, (1992)] |
|
On the role of the bridging dicyanamidobenzene ligand in a n...
2006-11-13 [Inorg. Chem. 45(23) , 9332-45, (2006)] |
Home | MSDS/SDS Database Search | Journals | Product Classification | Biologically Active Compounds | Selling Leads | About Us | Disclaimer
Copyright © 2026 ChemSrc All Rights Reserved