Chemical & Pharmaceutical Bulletin
2003-02-01
O-demethylation and sulfation of 7-methoxylated flavanones by Cunninghamella elegans.
Abdel-Rahim Sayed Ibrahim, Ahmed Mohamed Galal, Mohammed Shamim Ahmed, Gabir Salem Mossa
Index: Chem. Pharm. Bull. 51(2) , 203-6, (2003)
Full Text: HTML
Abstract
Metabolism of 7-O-methylnaringenin (sakuranetin) by Cunninghamella elegans NRRL 1392 yielded naringenin and naringenin-4'-sulfate. C. elegans also converted 5, 3', 4'-trihydroxy-7-methoxyflavanone into eriodictyol-4'-sulfate. Furthermore, incubation of 5, 4'-dihydroxy-7, 3'-dimethoxyflavanone with the same fungus gave homoeriodictyol (5, 7, 4'-trihydroxy-3'-methoxyflavanone) and homoeriodicytol-7-sulfate. The structures of the new metabolites were established by spectral analysis including 2D-NMR, HR-ESI-FT-MS beside hydrolysis by acid.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
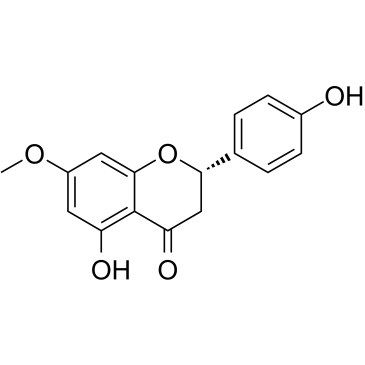 |
Sakuranetin
CAS:2957-21-3 |
C16H14O5 |
Related Articles:
More...
|
A nicotinic receptor-mediated anti-inflammatory effect of th...
2014-10-01 [Fitoterapia 98 , 11-21, (2014)] |
|
In vitro antileishmanial and antitrypanosomal activities of ...
2012-02-01 [Exp. Parasitol. 130(2) , 141-5, (2012)] |
|
Isoprenylated flavonoid and adipogenesis-promoting constitue...
2012-04-27 [J. Nat. Prod. 75(4) , 699-706, (2012)] |
|
Induced volatiles in elicitor-treated and rice blast fungus-...
2002-12-01 [Biosci. Biotechnol. Biochem. 66(12) , 2549-59, (2002)] |
|
Sakuranetin induces adipogenesis of 3T3-L1 cells through enh...
2008-01-01 [Biochem. Biophys. Res. Commun. 372(4) , 835-9, (2008)] |