| Structure | Name/CAS No. | Articles |
|---|---|---|
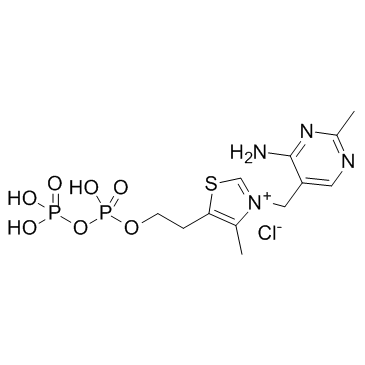 |
Cocarboxylase
CAS:154-87-0 |
|
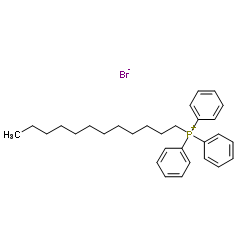 |
Dodecyl(triphenyl)phosphonium bromide
CAS:15510-55-1 |