Translocation versus cyclisation in radicals derived from N-3-alkenyl trichloroacetamides.
M Luisa Marin, Ramon J Zaragoza, Miguel A Miranda, Faïza Diaba, Josep Bonjoch
Index: Org. Biomol. Chem. 9(9) , 3180-7, (2011)
Full Text: HTML
Abstract
Under radical reaction conditions, two different and competitive reaction pathways were observed for N-(α-methylbenzyl)trichloroacetamides with a N-3-cyclohexenyl substituent: 1,4-hydrogen translocation and radical addition to a double bond. However, for radicals with an acyclic alkenyl side chain, the direct cyclisation process was exclusively observed. The dichotomy between translocation and direct radical cyclisation in these substrates has been theoretically studied using density functional theory (DFT) methods at the B3LYP/6-31G** computational level.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
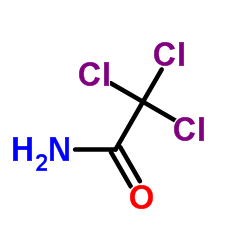 |
2,2,2-Trichloroacetamide
CAS:594-65-0 |
C2H2Cl3NO |
|
An improved synthesis of (-)-5,11-dideoxytetrodotoxin.
2013-02-15 [J. Org. Chem. 78(4) , 1699-705, (2013)] |
|
Activation of glycosyl trichloroacetimidates with perchloric...
2010-09-23 [Carbohydr. Res. 345(14) , 2074-8, (2010)] |
|
Synthetic studies toward the anthrax tetrasaccharide: altern...
2012-07-15 [Carbohydr. Res. 356 , 115-31, (2012)] |
|
Stereoselective rearrangement of trichloroacetimidates: appl...
2014-01-01 [Org. Lett. 11(11) , 2433-6, (2009)] |
|
Rhodium-catalyzed regio- and enantioselective amination of r...
2012-12-07 [Chem. Commun. (Camb.) 48(94) , 11531-3, (2012)] |