Synthetic studies toward the anthrax tetrasaccharide: alternative synthesis of this antigen
Ophélie Milhomme, Sandrine G.Y. Dhénin, Florence Djedaïni-Pilard, Vincent Moreau, Cyrille Grandjean
Index: Carbohydr. Res. 356 , 115-31, (2012)
Full Text: HTML
Abstract
The synthesis of the anthrax tetrasaccharide, amenable for conjugation, has been envisaged by both [2+2] and [1+3] approaches from D-fucose and L-rhamnose. The successful route reported herein relies on a [1+3] strategy in which the 1,2-trans-glycosidic linkages have been secured using a participating group at the 2-position of the donors using conventional thio as well as trichloroacetimidate glycosylation chemistry. The exchange of the ester to benzyl protective groups on the rhamnosyl moiety was key to achieve the final assembly and functionalization of the tetrasaccharide.Copyright © 2012 Elsevier Ltd. All rights reserved.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
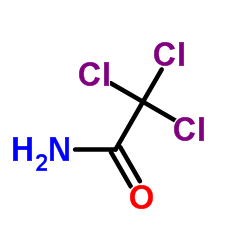 |
2,2,2-Trichloroacetamide
CAS:594-65-0 |
C2H2Cl3NO |
|
An improved synthesis of (-)-5,11-dideoxytetrodotoxin.
2013-02-15 [J. Org. Chem. 78(4) , 1699-705, (2013)] |
|
Activation of glycosyl trichloroacetimidates with perchloric...
2010-09-23 [Carbohydr. Res. 345(14) , 2074-8, (2010)] |
|
Stereoselective rearrangement of trichloroacetimidates: appl...
2014-01-01 [Org. Lett. 11(11) , 2433-6, (2009)] |
|
Rhodium-catalyzed regio- and enantioselective amination of r...
2012-12-07 [Chem. Commun. (Camb.) 48(94) , 11531-3, (2012)] |
|
Influence of the solvent in low temperature glycosylations w...
2011-09-06 [Carbohydr. Res. 346(12) , 1495-502, (2011)] |