QM/MM study of the mechanism of enzymatic limonene 1,2-epoxide hydrolysis.
Q Q Hou, X Sheng, J H Wang, Y J Liu, C B Liu
Index: Biochim. Biophys. Acta 1824(2) , 263-8, (2012)
Full Text: HTML
Abstract
Limonene 1,2-epoxide hydrolase (LEH) is completely different from those of classic epoxide hydrolases (EHs) which catalyze the hydrolysis of epoxides to vicinal diols. A novel concerted general acid catalysis step involving the Asp101-Arg99-Asp132 triad is proposed to play an important role in the mechanism. Combined quantum-mechanical/molecular-mechanical (QM/MM) calculations gave activation barriers of 16.9 and 25.1kcal/mol at the B3LYP/6-31G(d,p)//CHARMM level for nucleophilic attack on the more and less substituted epoxide carbons, respectively. Furthermore, the important roles of residues Arg99, Tyr53 and Asn55 on mutated LEH were evaluated by QM/MM-scanned energy mapping. These results may provide an explanation for site-directed mutagenesis.Copyright © 2011 Elsevier B.V. All rights reserved.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
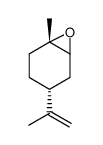 |
(+)-Trans-limonene1,2-epoxide
CAS:6909-30-4 |
C10H16O |
|
Catalytic mechanism of limonene epoxide hydrolase, a theoret...
2005-10-19 [J. Am. Chem. Soc. 127(41) , 14339-47, (2005)] |
|
Total synthesis of (+)-cymbodiacetal: a re-evaluation of the...
2010-12-17 [J. Org. Chem. 75(24) , 8465-70, (2010)] |
|
Metabolism of limonene-1,2-epoxide in the rat.
1980-12-01 [Xenobiotica 10(12) , 859-61, (1980)] |
|
Indoloquinazoline alkaloids from Euodia rutaecarpa and their...
2011-10-01 [J. Asian Nat. Prod. Res. 13(11) , 977-83, (2011)] |
|
The structures of alpha 2u-globulin and its complex with a h...
1999-04-01 [Acta Crystallogr. D Biol. Crystallogr. 55(Pt 4) , 753-62, (1999)] |