| Structure | Name/CAS No. | Articles |
|---|---|---|
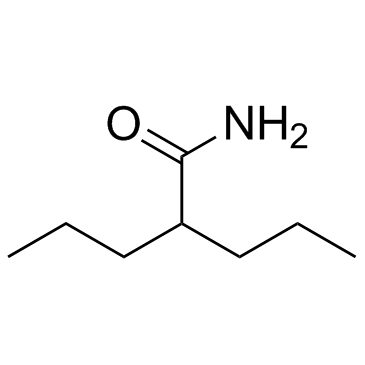 |
Valpromide
CAS:2430-27-5 |
|
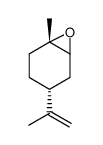 |
(+)-Trans-limonene1,2-epoxide
CAS:6909-30-4 |