| Structure | Name/CAS No. | Articles |
|---|---|---|
 |
N-hexane
CAS:110-54-3 |
|
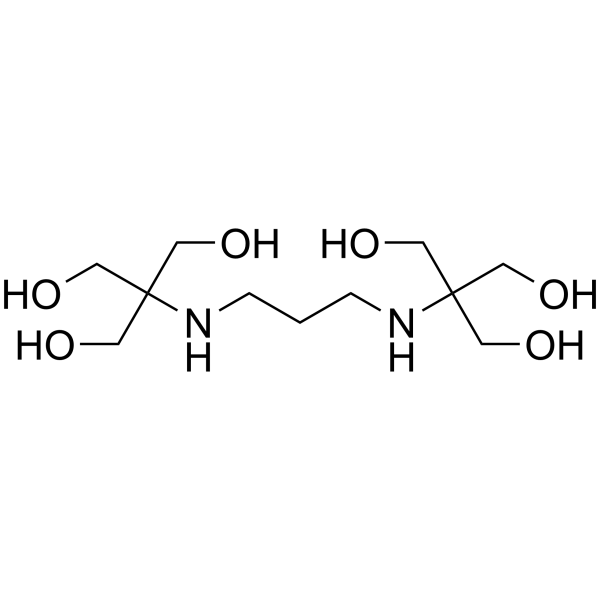 |
BIS-TRIS PROPANE
CAS:64431-96-5 |
|
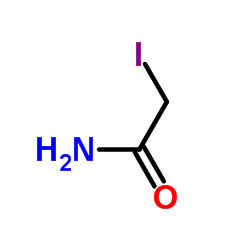 |
Iodoacetamide
CAS:144-48-9 |
|
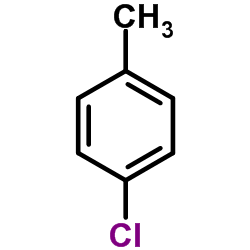 |
4-Chlorotoluene
CAS:106-43-4 |
|
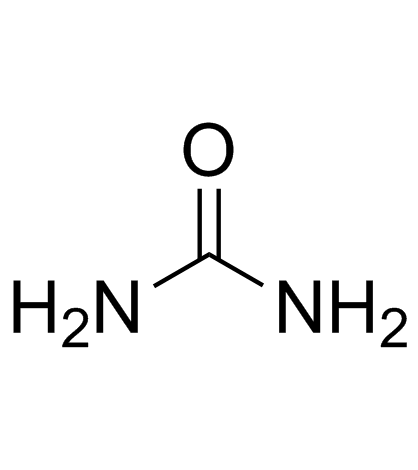 |
Urea
CAS:57-13-6 |