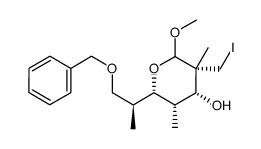
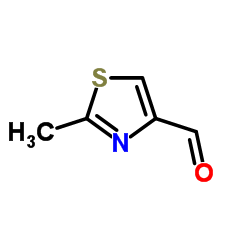
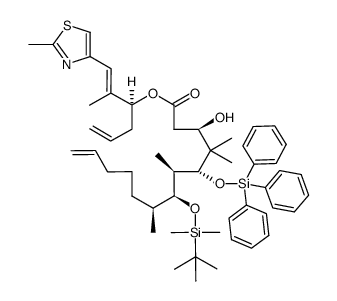
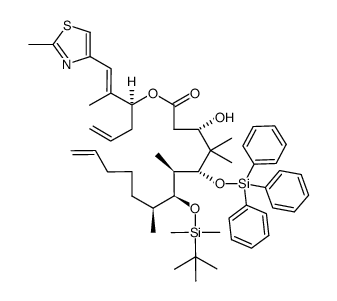
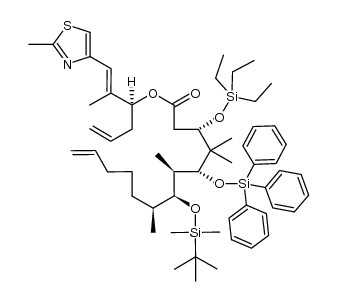
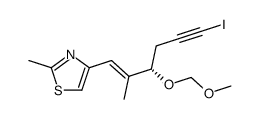
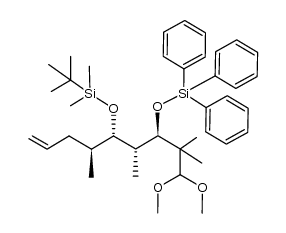
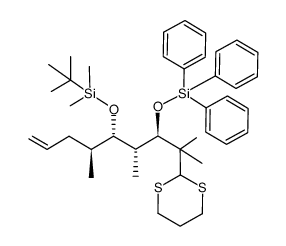
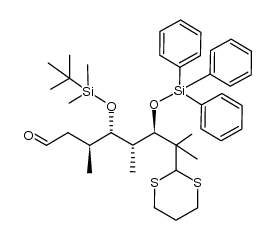
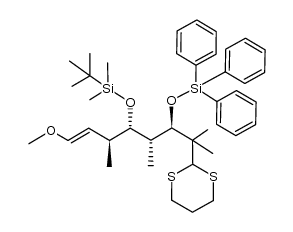
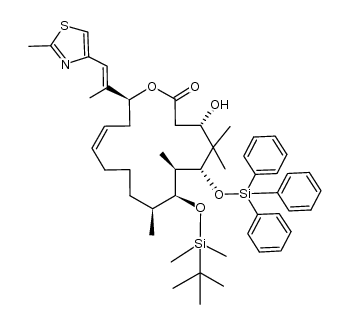
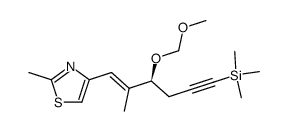
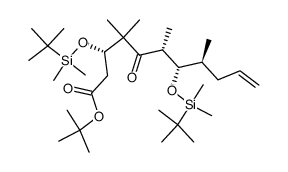
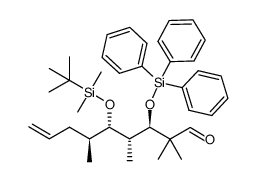
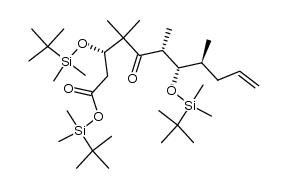
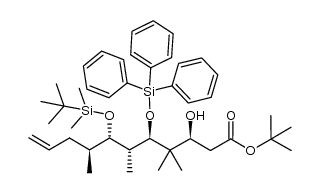
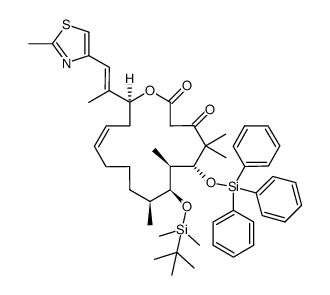
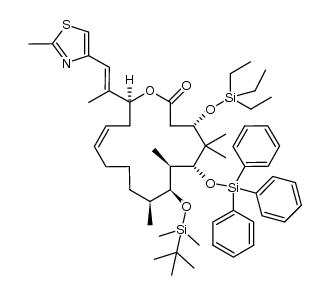
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |

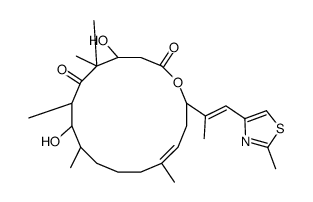
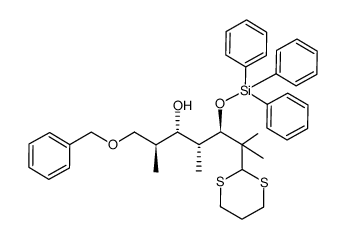

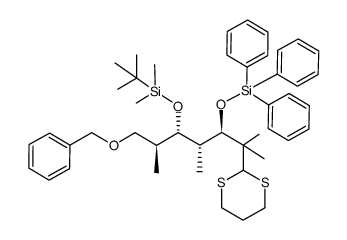
![(1E,3Z)-1-methoxy-2-methyl-3-[(trimethylsilyl)oxy]-penta-1,3-diene Structure](https://image.chemsrc.com/caspic/109/72486-93-2.png)
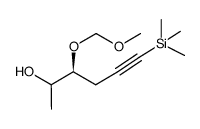
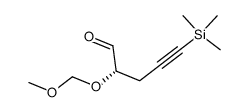
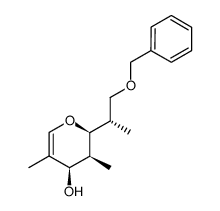
![(1S)-[(1E)-methyl-2-(2-methyl-1,3-thiazol-4-yl)ethynyl]-3-butenyl acetate Structure](https://image.chemsrc.com/caspic/246/188259-62-3.png)