| 结构式 | 名称/CAS号 | 全部文献 |
|---|---|---|
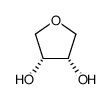 |
1,4-酐-赤藓糖醇
CAS:4358-64-9 |
|
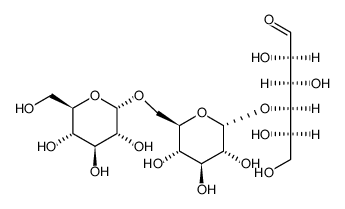 |
D-潘糖
CAS:33401-87-5 |
| 结构式 | 名称/CAS号 | 全部文献 |
|---|---|---|
 |
1,4-酐-赤藓糖醇
CAS:4358-64-9 |
|
 |
D-潘糖
CAS:33401-87-5 |