| 结构式 | 名称/CAS号 | 全部文献 |
|---|---|---|
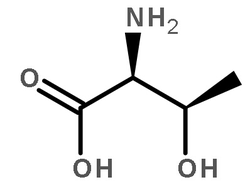 |
蛋白激酶A 来源于牛心脏
CAS:9026-43-1 |
|
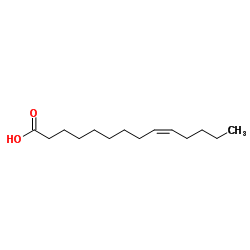 |
肉豆蔻油酸
CAS:544-64-9 |
| 结构式 | 名称/CAS号 | 全部文献 |
|---|---|---|
 |
蛋白激酶A 来源于牛心脏
CAS:9026-43-1 |
|
 |
肉豆蔻油酸
CAS:544-64-9 |