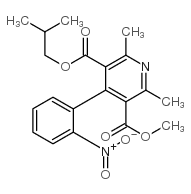
Nisoldipine

Nisoldipine structure
|
Common Name | Nisoldipine | ||
|---|---|---|---|---|
| CAS Number | 63675-72-9 | Molecular Weight | 388.414 | |
| Density | 1.2±0.1 g/cm3 | Boiling Point | 505.8±50.0 °C at 760 mmHg | |
| Molecular Formula | C20H24N2O6 | Melting Point | 147-148°C | |
| MSDS | Chinese USA | Flash Point | 259.7±30.1 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of NisoldipineNisoldipine(BAY-k 5552; Sular) is a calcium channel blocker belonging to the dihydropyridines class, specific for L-type Cav1.2 with IC50 of 10 nM. IC50 value: 10 nMTarget: L-type Cav1.2Nisoldipine is a potent blocker of L-type calcium channels. Nisoldipine binds directly to inactive calcium channels stabilizing their inactive conformation Similar to other DHP CCBs. Nisoldipine displays selectivity for arterial smooth muscle cells due to great number of inactive channels and the α1 subunit of the channel. Nisoldipine is about 30 times less selective for delayed-rectifier K+ channels than for L-type Ca2+ channels, which inhibits IKr (rapidly activating delayed-rectifier K+ current) with IC50 of 23 μM, and IKs (slowly activating delayed-rectifier K+ current)with IC50 of 40 μM in guinea-pig ventricular myocytes. Nisoldipine also displays antioxidant potency with IC50 of 28.2 μM both before and after the addition of active oxygen. |
| Name | methyl 2-methylpropyl 2,6-dimethyl-4-(2-nitrophenyl)-1,4-dihydropyridine-3,5-dicarboxylate |
|---|---|
| Synonym | More Synonyms |
| Description | Nisoldipine(BAY-k 5552; Sular) is a calcium channel blocker belonging to the dihydropyridines class, specific for L-type Cav1.2 with IC50 of 10 nM. IC50 value: 10 nMTarget: L-type Cav1.2Nisoldipine is a potent blocker of L-type calcium channels. Nisoldipine binds directly to inactive calcium channels stabilizing their inactive conformation Similar to other DHP CCBs. Nisoldipine displays selectivity for arterial smooth muscle cells due to great number of inactive channels and the α1 subunit of the channel. Nisoldipine is about 30 times less selective for delayed-rectifier K+ channels than for L-type Ca2+ channels, which inhibits IKr (rapidly activating delayed-rectifier K+ current) with IC50 of 23 μM, and IKs (slowly activating delayed-rectifier K+ current)with IC50 of 40 μM in guinea-pig ventricular myocytes. Nisoldipine also displays antioxidant potency with IC50 of 28.2 μM both before and after the addition of active oxygen. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 505.8±50.0 °C at 760 mmHg |
| Melting Point | 147-148°C |
| Molecular Formula | C20H24N2O6 |
| Molecular Weight | 388.414 |
| Flash Point | 259.7±30.1 °C |
| Exact Mass | 388.163422 |
| PSA | 110.45000 |
| LogP | 4.38 |
| Vapour Pressure | 0.0±1.3 mmHg at 25°C |
| Index of Refraction | 1.544 |
| Storage condition | -20°C Freezer |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
|
~52% 
Nisoldipine CAS#:63675-72-9 |
| Literature: ERREGIERRE, S.P.A. Patent: WO2004/2958 A1, 2004 ; Location in patent: Page 4-5 ; |
|
~% 
Nisoldipine CAS#:63675-72-9 |
| Literature: US5786344 A1, ; |
| HS Code | 2942000000 |
|---|
|
Fast disintegrating tablets of nisoldipine for intra-oral administration.
Pharm. Dev. Technol. 19(6) , 641-50, (2014) Nisoldipine is a calcium channel blocker with low and variable oral bioavailability. This was attributed to slow dissolution and presystemic metabolism. Accordingly, the objective of this work was to ... |
|
|
Anti-arrhythmic and hemodynamic effects of oxy nifedipine, oxy nimodipine, oxy nitrendipine and oxy nisoldipine.
Pharmacol. Res. 66(4) , 300-8, (2012) Our previous studies have established cardio-protective effects of furnidipine and its active metabolites. We therefore decided to compare the influence of oral and intravenous administration of furni... |
|
|
Why is nisoldipine a specific agent in ischemic left ventricular dysfunction?
Am. J. Cardiol. 75(13) , 36E-40E, (1995) Nisoldipine is a dihydropyridine calcium entry blocker that inhibits contraction of vascular smooth muscle with a potency that is 2-3 times greater than its impact on myocardial contractility. In isol... |
| methyl 2-methylpropyl 2,6-dimethyl-4-(2-nitrophenyl)-1,4-dihydropyridine-3,5-dicarboxylate |
| 3,5-Pyridinedicarboxylic acid, 1,4-dihydro-2,6-dimethyl-4-(2-nitrophenyl)-, methyl 2-methylpropyl ester |
| EINECS 264-407-7 |
| Nisoldipine |
| Isobutyl 1,4-Dihydro-5-methoxycarbonyl-2,6-dimethyl-4-(2-nitrophenyl)-3-pyridinecarboxylate |
| 3,5-Pyridinedicarboxylic acid, 1,4-dihydro-2,6-dimethyl-4-(3-nitrophenyl)-, methyl 2-methylpropyl ester |
| Isobutyl Methyl 1,4-Dihydro-2,6-dimethyl-4-(o-nitrophenyl)-3,5-pyridinedicarboxylate |
| MFCD00478055 |
| 3-O-methyl 5-O-(2-methylpropyl) 2,6-dimethyl-4-(2-nitrophenyl)-1,4-dihydropyridine-3,5-dicarboxylate |
| Isobutyl methyl 2,6-dimethyl-4-(2-nitrophenyl)-1,4-dihydropyridine-3,5-dicarboxylate |
| 2,6-Dimethyl-3-carbomethoxy-4-(2-nitrophenyl)-5-carbisobutoxy-1,4-dihydropyridine |
| Baymycard |
| Isobutyl methyl 2,6-dimethyl-4-(3-nitrophenyl)-1,4-dihydro-3,5-pyridinedicarboxylate |
| UNII:4I8HAB65SZ |
| Syscor |
| 1,4-Dihydro-2,6-dimethyl-4-(2-nitrophenyl)-3,5-pyridinedicarboxylic Acid Methyl 2-Methylpropyl Ester |
| Zadipina |
| Isobutyl methyl 2,6-dimethyl-4-(2-nitrophenyl)-1,4-dihydro-3,5-pyridinedicarboxylate |
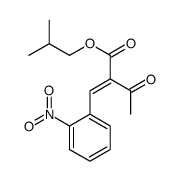

 CAS#:103026-83-1
CAS#:103026-83-1
