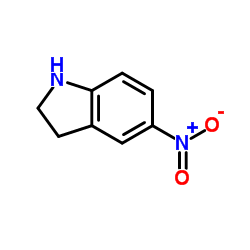
OAC-2
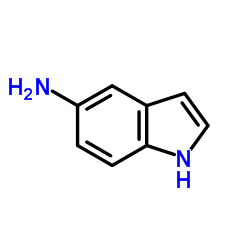
OAC-2 structure
|
Common Name | OAC-2 | ||
|---|---|---|---|---|
| CAS Number | 6019-39-2 | Molecular Weight | 236.269 | |
| Density | 1.3±0.1 g/cm3 | Boiling Point | 364.3±15.0 °C at 760 mmHg | |
| Molecular Formula | C15H12N2O | Melting Point | N/A | |
| MSDS | N/A | Flash Point | 174.1±20.4 °C | |
Use of OAC-2OAC2 is an Oct4-activating compound which activates expression through the Oct4 gene promoter. |
| Name | oac2 |
|---|---|
| Synonym | More Synonyms |
| Description | OAC2 is an Oct4-activating compound which activates expression through the Oct4 gene promoter. |
|---|---|
| Related Catalog | |
| In Vitro | Octamer-binding transcription factor 4 (Oct4) is a master regulator of the induction and maintenance of cellular pluripotency, and has crucial roles in early stages of differentiation. It is the only factor that cannot be substituted by other members of the same protein family to induce pluripotency[1]. Oct4 has been shown to be an essential regulator of embryonic stem cell (ESC) pluripotency and key to the reprogramming process. OAC2 is a structural analog of OAC1. OAC2 activates both Oct4 and Nanog reporters to a similar extent as OAC1. OAC1 and its two structural analogs OAC2 and OAC3 enhances reprogramming efficiency fourfold, up to as high as 2.75%, and accelerates the appearance of iPSC colonies 3 to 4 d when used in combination with the four reprogramming factors, Oct4, Sox2, Klf4, and c-Myc[2]. |
| Cell Assay | The Oct4-luc or Nanog-luc cells are treated with compound OAC1 or its structural analogs OAC2, OAC3 at 1 μM concentration or at indicated concentrations. Other compounds used include 2 μM BIO, 2 μM BIX, 2 μM 5'-azacytidine, 25 μg/mL Vitamin C, 10 nM Am580, 5 μM tranylcypromine, and 0.5 mM valporic acid. Luciferase reporter assays are performed 24 h after compound treatment or at indicated time points. For Topflash reporter assays, 0.2 μg β-catenin–responsive Topflash reporter gene plasmid is introduced into CV1 cells using trasfection. Compounds are added 6 h after transfection. Luciferase activity is measured 48 h after compound treatment using the Glo Luciferase Assay System[2]. |
| References |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Boiling Point | 364.3±15.0 °C at 760 mmHg |
| Molecular Formula | C15H12N2O |
| Molecular Weight | 236.269 |
| Flash Point | 174.1±20.4 °C |
| Exact Mass | 236.094955 |
| PSA | 44.89000 |
| LogP | 2.54 |
| Appearance of Characters | light yellow solid |
| Vapour Pressure | 0.0±0.8 mmHg at 25°C |
| Index of Refraction | 1.743 |
| Storage condition | -20℃ |
|
~% 
OAC-2 CAS#:6019-39-2 |
| Literature: ASTEX TECHNOLOGY LIMITED Patent: WO2003/87087 A2, 2003 ; Location in patent: Page/Page column 39 ; WO 03/087087 A2 |
|
~97% 
OAC-2 CAS#:6019-39-2 |
| Literature: Macor; Blank; Fox; Lebel; Newman; Post; Ryan; Schmidt; Schulz; Koe Journal of Medicinal Chemistry, 1994 , vol. 37, # 16 p. 2509 - 2512 |
|
~% 
OAC-2 CAS#:6019-39-2 |
| Literature: Terent'ew et al. Zhurnal Obshchei Khimii, 1959 , vol. 29, p. 2541,2550; engl. Ausg. S. 2504, 2511 |
|
~% 
OAC-2 CAS#:6019-39-2 |
| Literature: Terent'ew et al. Zhurnal Obshchei Khimii, 1959 , vol. 29, p. 2541,2550; engl. Ausg. S. 2504, 2511 |
|
~% 
OAC-2 CAS#:6019-39-2 |
| Literature: Terent'ew et al. Zhurnal Obshchei Khimii, 1959 , vol. 29, p. 2541,2550; engl. Ausg. S. 2504, 2511 |
|
~% 
OAC-2 CAS#:6019-39-2 |
| Literature: Terent'ew et al. Zhurnal Obshchei Khimii, 1959 , vol. 29, p. 2541,2550; engl. Ausg. S. 2504, 2511 |
| 3-Methyl-4-carboxy-5-benzamino-isothiazol |
| Benzamide, N-1H-indol-5-yl- |
| 5-Benzamidoindol |
| N-Indol-5-yl-benzamid |
| N-(1H-Indol-5-yl)benzamide |
| N-indol-5-yl-benzamide |
| 5-benzoylaminoindole |
| 3-Methyl-5-benzamido-isothiazol-4-carbonsaeure |
| 5-benzoylamino-3-methyl-isothiazole-4-carboxylic acid |
| 5-phenylcarbonylamino-1H-indole |
| 5-benzamidoindole |