Lithium chromate
Modify Date: 2024-01-11 19:56:16
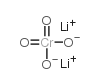
Lithium chromate structure
|
Common Name | Lithium chromate | ||
|---|---|---|---|---|
| CAS Number | 14307-35-8 | Molecular Weight | 129.87600 | |
| Density | 1.355 | Boiling Point | N/A | |
| Molecular Formula | CrLi2O4 | Melting Point | 844℃ | |
| MSDS | N/A | Flash Point | N/A | |
| Name | dilithium,dioxido(dioxo)chromium |
|---|---|
| Synonym | More Synonyms |
| Density | 1.355 |
|---|---|
| Melting Point | 844℃ |
| Molecular Formula | CrLi2O4 |
| Molecular Weight | 129.87600 |
| Exact Mass | 129.95200 |
| PSA | 80.26000 |
| Water Solubility | 94.6 g/L at 25 ºC |
|
Section 1: Product Identification Chemical Name:Lithium chromate, min. 95% CAS Registry Number:14307-35-8 Formula:Li2CrO4 EINECS Number:238-244-7 Chemical Family:chromate and dichromate salts Synonym:Dilithium chromate
Section 2: Composition and Information on Ingredients IngredientCAS NumberPercentACGIH (TWA)OSHA (PEL) Title Compound14307-35-8100%0.05mg/m31mg/10m3 Section 3: Hazards Identification Irritating to skin, eyes and respiratory tract. A recognized carcinogen. Lithium is toxic to the central nervous Emergency Overview: system and may damage kidneys and act as a teratogen. Primary Routes of Exposure:Ingestion, inhalation, skin and eyes Eye Contact:Causes irritatin of the eyes Skin Contact:causes irritation of the skin Inhalation:Causes inflammation and ulceration of the respiratory tract. Ingestion may lead to dizziness, abdominal cramps, vomiting, bloody diarrhea, weakness, shock, and Ingestion: convulsions. Acute Health Affects:Irritating to skin, eyes, and respiratory system. Contact with breaks in the skin can cause ulceration. Cancer hazard. May cause dermatitis, ulceration of the respiratory and gastrointestinal tract, acute hepatitis, Chronic Health Affects: erosion and discoloration of the teeth. Possible reproductive hazard. May cause heritable genetic damage. NTP:Yes IARC:Yes OSHA:No SECTION 4: First Aid Measures Immediately flush the eyes with copious amounts of water for at least 10-15 minutes. A victim may need Eye Exposure: assistance in keeping their eye lids open. Get immediate medical attention. Wash the affected area with water. Remove contaminated clothes if necessary. Seek medical assistance if Skin Exposure: irritation persists. Remove the victim to fresh air. Closely monitor the victim for signs of respiratory problems, such as difficulty Inhalation: in breathing, coughing, wheezing, or pain. In such cases seek immediate medical assistance. Seek medical attention immediately. Keep the victim calm. Give the victim water (only if conscious). Induce Ingestion: vomiting only if directed by medical personnel. SECTION 5: Fire Fighting Measures Flash Point:not applicable Autoignition Temperature:none Explosion Limits:none Extinguishing Medium:water, carbon dioxide, foam or dry powder If involved in a fire, fire fighters should be equipped with a NIOSH approved positive pressure self-contained Special Fire Fighting Procedures: breathing apparatus and full protective clothing. Hazardous Combustion andIf involved in a fire this material may emit toxic fumes of chromium oxides. Decomposion Products: Unusual Fire or Explosion Hazards: Strong oxidizer. May accelerate combustion of organics. SECTION 6: Accidental Release Measures Spill and Leak Procedures:Small spills can be mixed with vermiculite or sodium carbonate and swept up. SECTION 7: Handling and Storage Handling and Storage:Store in a sealed container. Keep away from heat, moisture, and direct sunlight. SECTION 8: Exposure Controls and Personal Protection Eye Protection:Always wear approved safety glasses when handling a chemical substance in the laboratory. Skin Protection:Wear appropriate chemical resistant gloves and protective clothing. Ventilation:Material may form a fine dust. If possible, handle the material in an efficient fume hood. If in form of fine dust and ventilation is not available a respirator should be worn. The use of respirators Respirator: requires a Respirator Protection Program to be in compliance with 29 CFR 1910.134. Ventilation:Material may form a fine dust. If possible, handle the material in an efficient fume hood. Additional Protection:No additional protection required. SECTION 9: Physical and Chemical Properties Color and Form:yellow xtl. Molecular Weight:129.87 Melting Point:no data Boiling Point:no data Vapor Pressure:not applicable Specific Gravity:no data Odor:none Solubility in Water:soluble SECTION 10: Stability and Reactivity Stability:air and moisture stable Hazardous Polymerization:no hazardous polymerization Conditions to Avoid:Contact with organics. Use good housekeeping practice to avoid accumulation of dust in work area. Incompatibility:reducing agents, organic matter, active metals, phosphorus and sulfur. Decomposition Products:none SECTION 11: Toxicological Information RTECS Data:No specific information available in the RTECS files. Carcinogenic Effects:Carcinogen (as Cr (VI) compound) Mutagenic Effects:Mutagen (as Cr (VI) compound) Tetratogenic Effects:No data available SECTION 12: Ecological Information Avoid release to the environment. Toxic to aquatic organisms. May cause long-term adverse effects in the Ecological Information: aquatic environment. SECTION 13: Disposal Considerations Disposal:Dispose of according to local, state and federal regulations. SECTION 14: Transportation Shipping Name (CFR):Oxidizing Solid, N.O.S. Hazard Class (CFR):5.1 Additional Hazard Class (CFR):NA Packaging Group (CFR):III UN ID Number (CFR):UN# 1479 Shipping Name (IATA):Oxidizing solid, N.O.S. Hazard Class (IATA):5.1 Additional Hazard Class (IATA):NA Packaging Group (IATA):III UN ID Number (IATA):UN# 1479 SECTION 15: Regulatory Information TSCA:Listed in the TSCA inventory. SARA (Title 313):Reportable under SARA 313. Second Ingredient:none SECTION 16 - ADDITIONAL INFORMATION N/A |
CHEMICAL IDENTIFICATION
|
| Hazard Codes | O,T+ |
|---|---|
| Risk Phrases | R8 |
| Safety Phrases | S17-S36/37/39-S53-S60 |
| RIDADR | 1479 |
| Packaging Group | III |
| Hazard Class | 6.1(b) |
| MFCD00016175 |
| LITHIUM CHROMATE |
| EINECS 238-244-7 |
| Dilithium chromate |
| Lithium Chromate dihydrate |
| Chromium lithium oxide |
| Chromic acid,dilithium salt |