2-nitro-5-aminobenzoic acid

2-nitro-5-aminobenzoic acid structure
|
Common Name | 2-nitro-5-aminobenzoic acid | ||
|---|---|---|---|---|
| CAS Number | 13280-60-9 | Molecular Weight | 182.133 | |
| Density | 1.6±0.1 g/cm3 | Boiling Point | 481.3±35.0 °C at 760 mmHg | |
| Molecular Formula | C7H6N2O4 | Melting Point | 234-242ºC | |
| MSDS | Chinese USA | Flash Point | 244.9±25.9 °C | |
| Name | 5-Amino-2-nitrobenzoic acid |
|---|---|
| Synonym | More Synonyms |
| Density | 1.6±0.1 g/cm3 |
|---|---|
| Boiling Point | 481.3±35.0 °C at 760 mmHg |
| Melting Point | 234-242ºC |
| Molecular Formula | C7H6N2O4 |
| Molecular Weight | 182.133 |
| Flash Point | 244.9±25.9 °C |
| Exact Mass | 182.032761 |
| PSA | 109.14000 |
| LogP | 1.17 |
| Vapour Pressure | 0.0±1.3 mmHg at 25°C |
| Index of Refraction | 1.682 |
| Storage condition | 2-8°C |
| Water Solubility | Insoluble |
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| Hazard Codes | Xn:Harmful; |
| Risk Phrases | R22;R36/37/38 |
| Safety Phrases | S26-S37/39 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| HS Code | 2922499990 |
| Precursor 8 | |
|---|---|
| DownStream 10 | |
| HS Code | 2922499990 |
|---|---|
| Summary | HS:2922499990 other amino-acids, other than those containing more than one kind of oxygen function, and their esters; salts thereof VAT:17.0% Tax rebate rate:9.0% Supervision conditions:AB(certificate of inspection for goods inward,certificate of inspection for goods outward) MFN tariff:6.5% General tariff:30.0% |
|
Positive cooperativity between acceptor and donor sites of the peptidoglycan glycosyltransferase.
Biochem. Pharmacol. 93(2) , 141-50, (2015) The glycosyltransferases of family 51 (GT51) catalyze the polymerization of lipid II to form linear glycan chains, which, after cross linking by the transpeptidases, form the net-like peptidoglycan ma... |
|
|
Pegylated fluorescent peptides as substrates of proteolytic enzymes.
Protein Pept. Lett. 19(12) , 1237-44, (2012) In this work the efficient and simple method of improvement specificity and solubility of low molecular weight proteinase substrates is described. The series of fluorescent substrates of selected prot... |
|
|
Sulfhydryl-based dendritic chain reaction.
Chem. Commun. (Camb.) 46(35) , 6575-7, (2010) A new dendritic chain reaction probe system was demonstrated to produce exponential signal amplification for the detection of sulfhydryl compounds. |
| EINECS 236-283-4 |
| MFCD00035748 |
| Benzoic acid, 5-amino-2-nitro- |
| 2-Nitro-5-aminobenzoic acid 3-Carboxy-4-nitroaniline |
| 5-Amino-2-nitrobenzoic acid |
| 2-nitro-5-aminobenzoic acid |
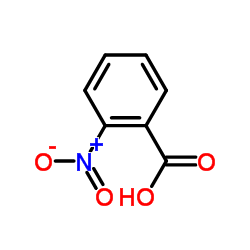 CAS#:552-16-9
CAS#:552-16-9 CAS#:1055310-21-8
CAS#:1055310-21-8 CAS#:1055310-22-9
CAS#:1055310-22-9 CAS#:1055310-23-0
CAS#:1055310-23-0 CAS#:1055310-24-1
CAS#:1055310-24-1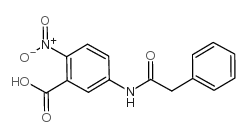 CAS#:52033-70-2
CAS#:52033-70-2 CAS#:4368-83-6
CAS#:4368-83-6 CAS#:20632-43-3
CAS#:20632-43-3 CAS#:50670-83-2
CAS#:50670-83-2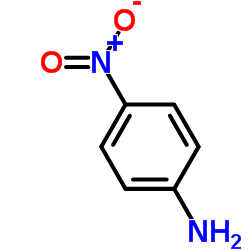 CAS#:100-01-6
CAS#:100-01-6 CAS#:106-50-3
CAS#:106-50-3 CAS#:610-74-2
CAS#:610-74-2 CAS#:610-37-7
CAS#:610-37-7 CAS#:35674-28-3
CAS#:35674-28-3 CAS#:35998-96-0
CAS#:35998-96-0 CAS#:77376-03-5
CAS#:77376-03-5![2-nitro-5-[(2,2,2-trifluoroacetyl)amino]benzoic acid structure](https://image.chemsrc.com/caspic/339/89165-27-5.png) CAS#:89165-27-5
CAS#:89165-27-5
