LY255283
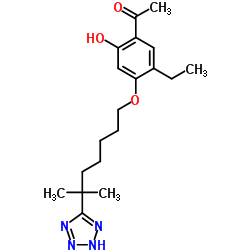
LY255283 structure
|
Common Name | LY255283 | ||
|---|---|---|---|---|
| CAS Number | 117690-79-6 | Molecular Weight | 360.451 | |
| Density | 1.2±0.1 g/cm3 | Boiling Point | 573.4±60.0 °C at 760 mmHg | |
| Molecular Formula | C19H28N4O3 | Melting Point | 160-162 °C | |
| MSDS | USA | Flash Point | 300.6±32.9 °C | |
Use of LY255283LY255283 is a LTB4 receptor (BLT2) antagonist, with an IC50 of ~100 nM for [3H]LTB4 binding to guinea pig lung membranes[1][2][3][4]. |
| Name | 1-[5-ethyl-2-hydroxy-4-[6-methyl-6-(2H-tetrazol-5-yl)heptoxy]phenyl]ethanone |
|---|---|
| Synonym | More Synonyms |
| Description | LY255283 is a LTB4 receptor (BLT2) antagonist, with an IC50 of ~100 nM for [3H]LTB4 binding to guinea pig lung membranes[1][2][3][4]. |
|---|---|
| Related Catalog | |
| Target |
LTB4:~100 nM (IC50, LTB4 binds to guinea pig lung membranes) |
| In Vitro | LY255283 compctitively reduces conlractilc responses of lung parcnchyma to LTB, (pA2 = 7.2)[2]. LY255283 (10 μM, 7 days) significantly suppresses the invasiveness of highly aggressive 253 J-BV bladder cancer cells[4]. Cell Viability Assay[4] Cell Line: 253 J-BV cells. Concentration: 5 or 10 μM. Incubation Time: 7 days. Result: Inhibition of BLT2 signaling attenuates aggressive migration by 253 J-BV cells. |
| In Vivo | LY255283 (3, 30 mg/kg) ameliorated lipopolysaccharide-induced ARDS in pigs, possibly by blocking the recruitment of activated PMNs into alveoli in a dose-dependent fashion[3]. LY255283 (2.5 mg/kg, ip) inhibits transitional cell carcinoma metastasis in mice models. The result suggests that a BLT2–Nox–ROS–NF–κB cascade plays a critical role in bladder cancer invasion and metastasis[4]. Animal Model: Mice (253 J-BV cells injected)[4]. Dosage: 2.5 mg/kg. Administration: IP injected 3 and 5 days after injection of cells. Result: By 12 weeks after injection, in mice treated with LY255283 only 0-3 nodules formed per lung, and histological analysis confirmed that the number of micrometastatic lesions was markedly reduced. |
| References |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 573.4±60.0 °C at 760 mmHg |
| Melting Point | 160-162 °C |
| Molecular Formula | C19H28N4O3 |
| Molecular Weight | 360.451 |
| Flash Point | 300.6±32.9 °C |
| Exact Mass | 360.216156 |
| PSA | 100.99000 |
| LogP | 4.04 |
| Vapour Pressure | 0.0±1.6 mmHg at 25°C |
| Index of Refraction | 1.553 |
| Hazard Codes | F+ |
|---|---|
| RIDADR | NONH for all modes of transport |
| HS Code | 2933990090 |
| HS Code | 2933990090 |
|---|---|
| Summary | 2933990090. heterocyclic compounds with nitrogen hetero-atom(s) only. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Leukotriene B4 Inhibits L-Type Calcium Channels via p38 Signaling Pathway in Vascular Smooth Muscle Cells.
Cell Physiol. Biochem. 37 , 1903-13, (2015) Arachidonic acid (AA) and its metabolites are important endogenous lipid messengers. In this study, we test the effect of Leukotriene B4 (LTB4), a 5-lipoxygenase metabolite of AA, on L-type calcium ch... |
|
|
12-Hydroxyheptadecatrienoic acid promotes epidermal wound healing by accelerating keratinocyte migration via the BLT2 receptor.
J. Exp. Med. 211(6) , 1063-78, (2014) Leukotriene B4 (LTB4) receptor type 2 (BLT2) is a G protein-coupled receptor (GPCR) for 12(S)-hydroxyheptadeca-5Z,8E,10E-trienoic acid (12-HHT) and LTB4. Despite the well-defined proinflammatory roles... |
|
|
A leukotriene B4 receptor-2 is associated with paclitaxel resistance in MCF-7/DOX breast cancer cells.
Br. J. Cancer 109(2) , 351-9, (2013) Breast cancer is the most common malignancy in women. Although chemotherapeutic agents, such as paclitaxel, are effective treatments for the majority of breast cancer patients, recurrence is frequent ... |
| 1-(5-Ethyl-2-hydroxy-4-(6-methyl-6-(1H-tetrazol-5-yl)heptyloxy)phenyl)ethanone |
| 1-(5-Ethyl-2-hydroxy-4-{[6-methyl-6-(2H-tetrazol-5-yl)heptyl]oxy}phenyl)ethanone |
| Ethanone, 1-[5-ethyl-2-hydroxy-4-[[6-methyl-6-(2H-tetrazol-5-yl)heptyl]oxy]phenyl]- |