288262-96-4
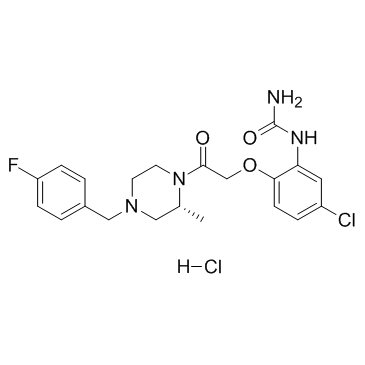
| Name | [5-chloro-2-[2-[(2R)-4-[(4-fluorophenyl)methyl]-2-methylpiperazin-1-yl]-2-oxoethoxy]phenyl]urea,hydrochloride |
|---|---|
| Synonyms |
BX471 (hydrochloride)
BX-471 UNII-L350B5LO1J |
| Description | BX471 hydrochloride (ZK-811752 hydrochloride) is a potent, selective non-peptide CCR1 antagonist with Ki of 1 nM for human CCR1, and exhibits 250-fold selectivity for CCR1 over CCR2, CCR5 and CXCR4. |
|---|---|
| Related Catalog | |
| Target |
MIP-1α-CCR1:1 nM (Ki) RANTES-CCR1:2.8 nM (Ki) MCP-3-CCR1:5.5 nM (Ki) |
| In Vitro | BX471 is a potent functional antagonist based on its ability to inhibit a number of CCR1-mediated effects including Ca2+ mobilization, increase in extracellular acidification rate, CD11b expression, and leukocyte migration. BX471 demonstrats a greater than 10,000-fold selectivity for CCR1 compared with 28 G-protein-coupled receptors[1]. BX471 is also able to displace 125I-MIP-1α/CCL3 binding to mouse CCR1 in a concentration-dependent manner with a Ki of 215±46 nM. Increasing concentrations of BX471 inhibits the Ca2+ transients induced by MIP-1α/CCL3 in both human and mouse CCR1 with IC50 of 5.8±1 nM and 198±7 nM, respectively[2]. BX471 (0.1-10 μM) shows a dose-dependent inhibition of RANTES-mediated and shear-resistant adhesion on IL-1β-activated microvascular endothelium in shear flow in isolated blood monocytes. BX471 also inhibits the RANTES-mediated adhesion of T lymphocytes to activated endothelium[4]. |
| In Vivo | BX471 (4 mg/kg, p.o. or i.v.) is orally active with a bioavailability of 60% in dogs. Furthermore, BX471 effectively reduces disease in a rat experimental allergic encephalomyelitis model of multiple sclerosis[1]. BX471 (20 mg/kg, s.c.) reaches peak plasma levels of 9 μM by around 30 minutes, and this rapidly declines to approximately 0.4 μM after 2 hours. From 4 to 8 hours the drug plasma levels drops to 0.1 μM or lower. Mice treated with 20 mg/kg of BX471 for 10 days shows a reduction of interstitial CD45 positive leukocytes of approximately 55%. BX471 has a borderline significant effect on the number of CCR5-positive CD8 cells in the peripheral blood. BX471 reduces the amount of FSP1-positive cells by 65% in UUO kidneys as compared with vehicle control[2]. Pretreatment witih BX471 reduces macrophage and neutrophil accumulation in kidney after ischemia-reperfusion injury[3]. |
| Cell Assay | Briefly, dermal microvascular endothelial cells grown to confluence in Petri dishes are stimulated with IL-1β (10 ng/mL) for 12 h followed by pre-incubation with RANTES (10 nM) for 30 min at 37°C just prior to assay. The plates are assembled as the lower wall in a parallel wall flow chamber and mounted on the stage of an Olympus IMT-2 inverted microscope with ×20 and ×40 phase-contrast objectives. Isolated human blood monocytes are isolated and resuspended at 5×105 cells/mL in assay buffer (HBSS) containing 10 mM HEPES, pH 7.4 and 0.5% human serum albumin. Shortly before the assay, 1 mM Mg2+ and 1 mM Ca2+ are added. The cell suspensions are kept in a heating block at 37°C during the assay and perfused into the flow chamber at a rate of 1.5 dyn/cm2 for 5 min. For inhibition experiments, monocytes are preincubated with BX471 at different concentrations (0.1-10 μM) or a Me2SO control for 10 min at 37°C. The number of firmLy adherent cells after 5 min is quantitated in multiple fields (at least five per experiment) by analysis of images recorded with a long integration JVC 3CCD video camera and a JVC SR L 900 E video recorder and are expressed as cells/mm2. The type of adhesion analyzed is restricted to primary, i.e. direct interactions of monocytes with endothelium. |
| Animal Admin | Fasted male beagle dogs (n=3 per treatment group) are given BX471 either by oral gavage or by intravenous injection via the cephalic vein at a dose of 4 mg/kg. The compound is dissolved in a vehicle of 40% aqueous cyclodextrin. Serial blood samples are collected utilizing an in-dwelling catheter in the jugular vein at the indicated time points up to 6 h post-dosing. EDTA is used as an anticoagulant. The samples are centrifuged (1000× g for 10 min at 4°C), and plasma is stored frozen until analyzed for drug levels by HPLC-MS (electrospray mode operated under a positive ion mode). Plasma samples are thawed and denatured by the addition of four parts of ice-cold methanol containing a fixed amount of an internal standard to one part of plasma. The resulting protein precipitate is removed by centrifugation at 5000× g, and the supernatants are analyzed directly. Concurrently plasma calibration standards of BX471 are prepared over the range of quantification, processed, and analyzed under identical conditions. A FISONS, VG Platform single quadrupole instrument is used in these analyses with an electrospray inlet operated at 3.57 kV. Chromatographic separation is accomplished using a YMC AQ octadecyl silane reversed phase column (4.6×250 mm) following a short isocratic elution method (35% methanol, 65% water containing 0.1% trifluoroacetic acid). The total column flow (1 mL/min) is split post-column to infuse 50 μL/min into the mass spectrometer. The chromatograms are collected over a total run time of 7.5 min/sample following a 50-μL injection on the column. The ions are collected in a single ion positive ionization mode. A calibration curve for quantification is generated by plotting ion current ratios between the internal standard peak and the analyte in the plasma standards over the quantification range. Calculations of percent oral availability is deduced from the area under curve measurements. Pharmacokinetic parameters are calculated using WinNonLin version 3.0. |
| References |
| Molecular Formula | C21H25Cl2FN4O3 |
|---|---|
| Molecular Weight | 471.35300 |
| Exact Mass | 470.12900 |
| PSA | 88.89000 |
| LogP | 4.34600 |
| Storage condition | 2-8℃ |