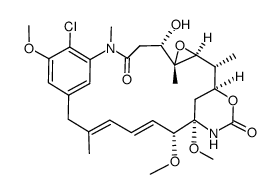
35846-53-8
| Name | maytansine |
|---|---|
| Synonyms |
maitansine
(3E,5E,7R,84S)-12c-(N-acetyl-N-methyl-L-alanyloxy)-14-chloro-10t,11c-epoxy-84-hydroxy-15,7r-dimethoxy-3,9c,11t,15-tetramethyl-(84r'H,86c'H)-15-aza-1(1,3)-benzena-8(4,6)-[1,3]oxazinana-cyclopentadecaphane-3,5-diene-82,14-dione Maytansine |
| Description | Maytansine is a highly potent microtubule-targeted compound that induces mitotic arrest and kills tumor cells at subnanomolar concentrations[1]. |
|---|---|
| Related Catalog | |
| In Vitro | Maytansine, at 6x10-8 M, irreversibly inhibits cell division in eggs of sea urchins and clams. Maytansine causes the disappearance of a mitotic apparatus or prevents one from forming if added at early stages. Maytansine does inhibit in vitro polymerization of tubulin. Maytansine inhibits in vitro polymerization of tubulin[2]. |
| References |
| Density | 1.32g/cm3 |
|---|---|
| Boiling Point | 895.1ºC at 760mmHg |
| Molecular Formula | C34H46ClN3O10 |
| Molecular Weight | 692.19600 |
| Flash Point | 495.1ºC |
| Exact Mass | 691.28700 |
| PSA | 156.47000 |
| LogP | 3.92820 |
| Index of Refraction | 1.59 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
|
~95% 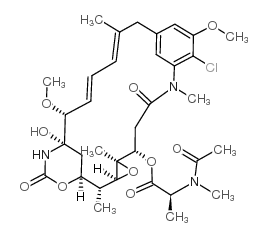
35846-53-8 |
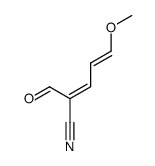
| Literature: Corey; Weigel; Chamberlin; et al. Journal of the American Chemical Society, 1980 , vol. 102, # 21 p. 6613 - 6615 |
|
~% 
35846-53-8 |
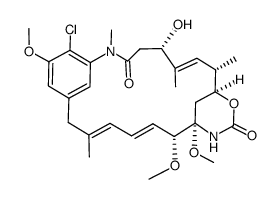
| Literature: Journal of the American Chemical Society, , vol. 102, # 21 p. 6613 - 6615 |
|
~% 
35846-53-8 |
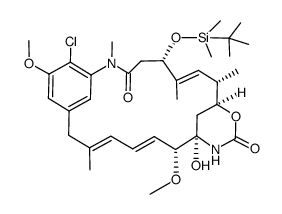
| Literature: Journal of the American Chemical Society, , vol. 102, # 21 p. 6613 - 6615 |
|
~% 
35846-53-8 |
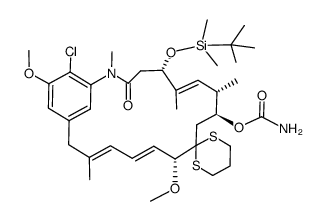
| Literature: Journal of the American Chemical Society, , vol. 102, # 21 p. 6613 - 6615 |
|
~% 
35846-53-8 |
| Literature: Journal of the American Chemical Society, , vol. 102, # 21 p. 6613 - 6615 |
|
~% 
35846-53-8 |
| Literature: Journal of the American Chemical Society, , vol. 102, # 21 p. 6613 - 6615 |
|
~% 
35846-53-8 |
| Literature: Journal of the American Chemical Society, , vol. 102, # 21 p. 6613 - 6615 |
| Precursor 5 | |
|---|---|
| DownStream 0 | |