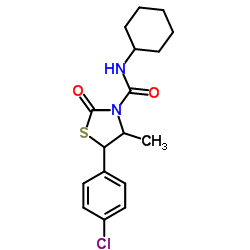
78587-05-0
| Name | (S,S)-hexythiazox |
|---|---|
| Synonyms |
Hexygon,Acariflor,Calibre
Savey Matacar Nissorun Acarflor Acariflor Trevi (4S,5S)-5-(4-chlorophenyl)-N-cyclohexyl-4-methyl-2-oxo-1,3-thiazolidine-3-carboxamide Calibre MFCD00137385 5-(4-Chlorophenyl)-N-cyclohexyl-4-methyl-2-oxo-1,3-thiazolidine-3-carboxamide Hexythiazox rac-(4R,5R)-5-(4-chlorophenyl)-N-cyclohexyl-4-methyl-2-oxo-1,3-thiazolidine-3-carboxamide (4R,5R)-rel-5-(4-chlorophenyl)-N-cyclohexyl-4-methyl-2-oxo-3-thiazolidinecarboxamide Ordoval Cesar (4RS,5RS)-5-(4-Chlorophenyl)-N-cyclohexyl-4-methyl-2-oxo-1,3-thiazolidine-3-carboxamide |
| Description | Hexythiazox is a selective acaricide with ovicidal, larvicidal and nymphicidal activities. Hexythiazox is widely used for chemical control of mites on cotton, fruits and vegetables. Hexythiazox is harmless to mammals and has no effect on beneficial insects and predators of mites[1][2]. |
|---|---|
| Related Catalog | |
| In Vitro | Hexythiazox is dissipated easily in strawberries, with the half-life ranging from 3.43 to 3.59 days[1]. Hexythiazox induces toxicity in larvae of the two-spotted spider mite, Tetranychus uritcae, and the European red mite, Panonychus ulmi with LC50 values of 0.15-0.58 mg AI/L and 0.23-0.62 mg AI/L, respectively[2]. |
| References |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Melting Point | 108-108.5°C |
| Molecular Formula | C17H21ClN2O2S |
| Molecular Weight | 352.879 |
| Flash Point | 100 °C |
| Exact Mass | 352.101227 |
| PSA | 74.71000 |
| LogP | 3.41 |
| Index of Refraction | 1.621 |
| Storage condition | 0-6°C |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Symbol |

GHS09 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H410 |
| Precautionary Statements | P273-P501 |
| Personal Protective Equipment | Eyeshields;Faceshields;Gloves |
| Hazard Codes | N:Dangerousfortheenvironment; |
| Risk Phrases | R50/53 |
| Safety Phrases | S60-S61 |
| RIDADR | UN 3077 |
| RTECS | XJ5396000 |
| HS Code | 2934100011 |
| HS Code | 2934100011 |
|---|