34642-77-8
| Name | amoxicillin sodium |
|---|---|
| Synonyms |
Penamox Inj.
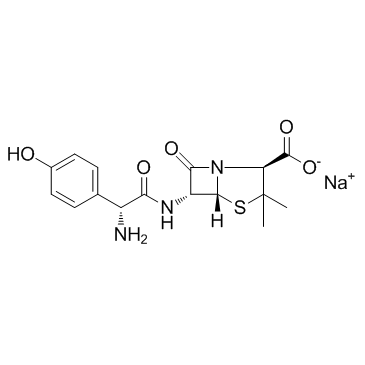
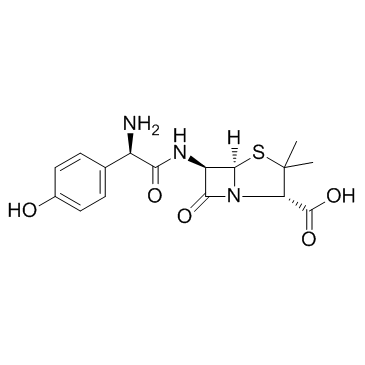
Sodium (2S,5R,6R)-6-{[(2R)-2-amino-2-(4-hydroxyphenyl)acetyl]amino}-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylate AmoxycillinSodiumSterileBp98 Amoxicillin sodium Amoxicillin (sodium) 4-Thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid, 6-[[(2R)-2-amino-2-(4-hydroxyphenyl)acetyl]amino]-3,3-dimethyl-7-oxo-, sodium salt, (2S,5R,6R)- (1:1) (2S,5R,6R)-6-[[(R)-Amino(4-hydroxyphenyl)acetyl]amino]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid sodium salt riotapen Acuotricina sodium amoxicillin Moxacin Inj. [2S-[2a,5a,6b[S*]]]-6-[[Amino[4-hydroxyphenyl]acetyl]amino]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic Acid Monosodium Salt AMOXYCILLIN SODIUM Monosodium [-]-[2S,5R,6R]-6-[[R]-2-Amino[4-hydroxyphenyl]acetamido]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylate MFCD08063910 EINECS 252-124-1 Novabritine Inj. AMoxicillin SodiuM+Clavulanate PotassiuM(4:1) Amoxycillin sodium salt Danoxillin amoxiclllln sodium Amoxycillin sodium Crystalline Sodium [2S-[2a,5a,6b[S*]]]-6-[[Amino[4-hydroxyphenyl]acetyl]amino]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylate Lamoxy Inj. |
| Description | Amoxicillin Sodium is a moderate- spectrum, bacteriolytic, β-lactam antibiotic.Target: AntibacterialAmoxicillin is a moderate-spectrum, bacteriolytic, β-lactam antibiotic in the aminopenicillin family used to treat bacterial infections caused by susceptible Gram-positive and Gram-negative microorganisms. It is usually the drug of choice within the class because it is better-absorbed, following oral administration, than other β-lactam antibiotics. Amoxicillin is susceptible to degradation by β-lactamase-producing bacteria, which are resistant to a narrow spectrum of β-lactam antibiotics, such as penicillin. For this reason, it is often combined with clavulanic acid, a β-lactamase inhibitor. This increases effectiveness by reducing its susceptibility to β-lactamase resistance. From Wikipedia. |
|---|---|
| Related Catalog | |
| References |
| Boiling Point | 743.2ºC at 760 mmHg |
|---|---|
| Molecular Formula | C16H18N3NaO5S |
| Molecular Weight | 387.386 |
| Exact Mass | 387.086487 |
| PSA | 161.09000 |
| HS Code | 2941109900 |
|---|
| Precursor 1 | |
|---|---|
| DownStream 0 | |
| HS Code | 2941109900 |
|---|