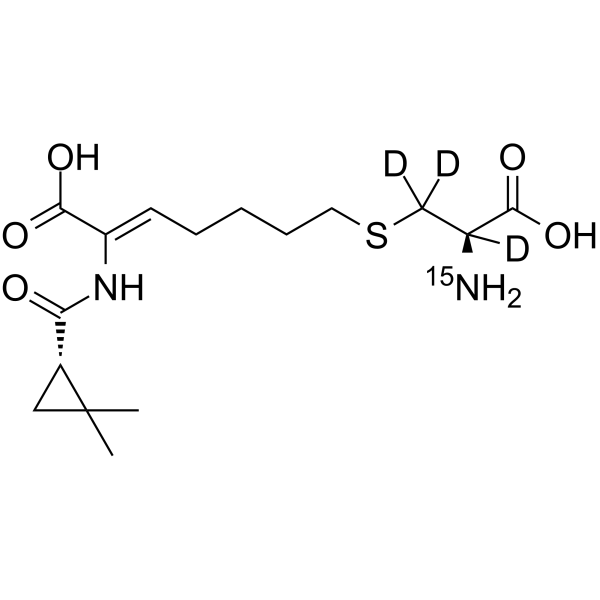
2738376-83-3
| Name | Cilastatin-15N,d3 |
|---|
| Description | Cilastatin-15N,d3 is a 15N-labeled and deuterium labeled Cilastatin (MK0791) is a reversible, competitive renal dehydropeptidase I inhibitor with an IC50 of 0.1 μM. Cilastatin inhibits the bacterial metallob-lactamase enzyme CphA with an IC50 of 178 μM. Cilastatin is an antibacterial adjunct[1][2][3]. |
|---|---|
| Related Catalog | |
| In Vitro | Stable heavy isotopes of hydrogen, carbon, and other elements have been incorporated into drug molecules, largely as tracers for quantitation during the drug development process. Deuteration has gained attention because of its potential to affect the pharmacokinetic and metabolic profiles of drugs[5]. |
| References |
| Molecular Formula | C16H23D3N15NO5S |
|---|---|
| Molecular Weight | 362.46 |