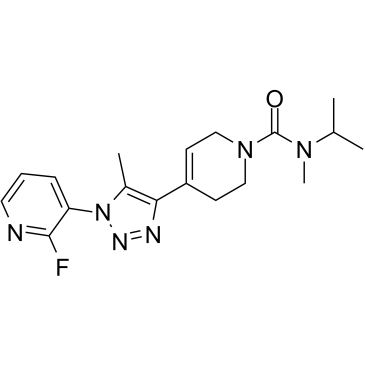
873551-53-2
| Name | FTIDC |
|---|---|
| Synonyms |
4-[1-(2-Fluoro-3-pyridinyl)-5-methyl-1H-1,2,3-triazol-4-yl]-N-isopropyl-N-methyl-3,6-dihydro-1(2H)-pyridinecarboxamide
FTIDC |
| Description | FTIDC is an orally active, noncompetitive, selective allosteric metabotropic glutamate receptor (mGluR) 1 antagonist with an IC50 of 5.8 nM for human mGluR1a. FTIDC has no species differences in its antagonistic activity on recombinant human, mouse, and rat mGluR1[1]. |
|---|---|
| Related Catalog | |
| Target |
mGluR1a:5.8 nM (IC50) mGluR5:6200 nM (IC50) |
| In Vitro | FTIDC inhibits L-glutamate-induced increases in intracellular Ca2+ concentrations, with IC50 values of 5.8 nM , 5.8 nM , 3.1 nM , 7.7 nM for human mGluR1a, rat mGluR1a, mouse mGluR1a, human mGluR1b in CHO cells, respectively[1]. |
| In Vivo | FTIDC (i.p. or p.o.; 1-30 mg/kg) reduces the duration of face-washing behavior elicited in a dosedependent manner and the inhibitory effect is statistically significant at 10 and 30 mg/kg with i.p. and 30 mg/kg with p.o.[1]. Animal Model: Male CD1 (ICR) mice of 6-weeks-old[1] Dosage: 1, 3, 10, and 30 mg/kg Administration: I.p. or p.o. Result: Reduced the duration of face-washing behavior elicited in a dosedependent manner and was statistically significant at 10 and 30 mg/kg with i.p. and 30 mg/kg with p.o.. |
| References |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Boiling Point | 558.2±60.0 °C at 760 mmHg |
| Molecular Formula | C18H23FN6O |
| Molecular Weight | 358.413 |
| Flash Point | 291.4±32.9 °C |
| Exact Mass | 358.191742 |
| LogP | 2.15 |
| Vapour Pressure | 0.0±1.5 mmHg at 25°C |
| Index of Refraction | 1.624 |