29307-03-7
| Name | Elephantopus carolinianus compd. B |
|---|---|
| Synonyms |
Deoxyelephantopin
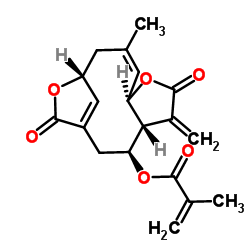
(3S,4R,8R,9Z,12R)-10-Methyl-5-methylene-6,14-dioxo-7,13-dioxatricyclo[10.2.1.0]pentadeca-1(15),9-dien-3-yl methacrylate 2-Propenoic acid, 2-methyl-, (3aR,4S,9R,11Z,12aR)-2,3,3a,4,5,9,10,12a-octahydro-11-methyl-3-methylene-2,7-dioxo-7H-9,6-methenofuro[2,3-f]oxacycloundecin-4-yl ester Isodeoxyelephantopin |
| Description | Deoxyelephantopin, a natural bioactive sesquiterpene lactone from Elephantopus scaber, has shown promising anticancer effects against a broad spectrum of cancers. Deoxyelephantopin inhibits NF-κB, MAPK, PI3K/Akt, and β-catenin signaling[1]. |
|---|---|
| Related Catalog | |
| Target |
NF-κB |
| In Vitro | Deoxyelephantopin inhibits the cell growth of HCT 116 (colorectal), K562 (chronic myeloid leukemia), KB (oral), and T47D (breast) cancer cell lines with IC50s of 7.46, 4.02, 3.35, 1.86 μg/mL, respectively[1]. Deoxyelephantopin increases the expression of p53, p-JNK, and p-p38 and decreases the expression of p-STAT3 and p-mTOR in cancer cells. Deoxyelephantopin downregulates MMP-2 and MMP-9, uPA, and uPAR mRNA levels in cancer cells[1]. |
| References |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Boiling Point | 584.3±50.0 °C at 760 mmHg |
| Melting Point | 150-153℃ |
| Molecular Formula | C19H20O6 |
| Molecular Weight | 344.358 |
| Flash Point | 258.1±30.2 °C |
| Exact Mass | 344.125977 |
| PSA | 78.90000 |
| LogP | 1.51 |
| Vapour Pressure | 0.0±1.6 mmHg at 25°C |
| Index of Refraction | 1.556 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATAMUTATION DATA
|
| Hazard Codes | Xi |
|---|