ML314
Modify Date: 2025-08-27 18:48:04
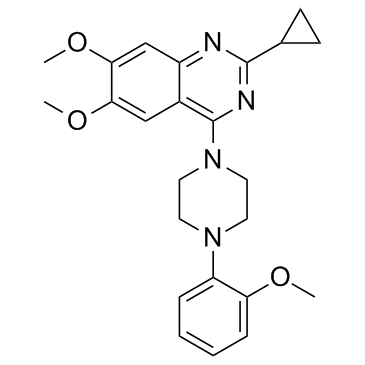
ML314 structure
|
Common Name | ML314 | ||
|---|---|---|---|---|
| CAS Number | 1448895-09-7 | Molecular Weight | 420.504 | |
| Density | 1.2±0.1 g/cm3 | Boiling Point | 496.1±45.0 °C at 760 mmHg | |
| Molecular Formula | C24H28N4O3 | Melting Point | N/A | |
| MSDS | N/A | Flash Point | 253.8±28.7 °C | |
Use of ML314ML314 is a potent molecule agonist of NTR1 (EC50 = 1.9 μM); showed good selectivity against NTR2 and GPR35, but did not stimulate Ca2+ mobilization.IC50 value: 1.9 uM (EC50) [1]Target: NTR1 agonistMedicinal chemistry optimization of MLS-0233108 led to ML314, the most potent molecule in this second series that exhibited full agonist behavior (100 %) on NTR1 (EC50 = 1.9 μM). ML314 showed good selectivity against NTR2 and GPR35, but did not stimulate Ca2+ mobilization. ML314 is potentially a biased agonist operating via the β-arrestin pathway rather than the traditional Gq coupled pathway. Signaling mediated by β-arrestin has distinct biochemical and functional consequences that may lead to physiological advantages as described below. This probe report describes the discovery and properties of ML301 and summarizes the HTS and follow-up campaign, which identified ML314. |
| Name | 2-cyclopropyl-6,7-dimethoxy-4-(4-(2-methoxyphenyl)piperazin-1-yl)quinazoline |
|---|---|
| Synonym | More Synonyms |
| Description | ML314 is a potent molecule agonist of NTR1 (EC50 = 1.9 μM); showed good selectivity against NTR2 and GPR35, but did not stimulate Ca2+ mobilization.IC50 value: 1.9 uM (EC50) [1]Target: NTR1 agonistMedicinal chemistry optimization of MLS-0233108 led to ML314, the most potent molecule in this second series that exhibited full agonist behavior (100 %) on NTR1 (EC50 = 1.9 μM). ML314 showed good selectivity against NTR2 and GPR35, but did not stimulate Ca2+ mobilization. ML314 is potentially a biased agonist operating via the β-arrestin pathway rather than the traditional Gq coupled pathway. Signaling mediated by β-arrestin has distinct biochemical and functional consequences that may lead to physiological advantages as described below. This probe report describes the discovery and properties of ML301 and summarizes the HTS and follow-up campaign, which identified ML314. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 496.1±45.0 °C at 760 mmHg |
| Molecular Formula | C24H28N4O3 |
| Molecular Weight | 420.504 |
| Flash Point | 253.8±28.7 °C |
| Exact Mass | 420.216156 |
| PSA | 59.95000 |
| LogP | 2.49 |
| Vapour Pressure | 0.0±1.3 mmHg at 25°C |
| Index of Refraction | 1.633 |
| InChIKey | SWEOAXMICIJCQC-UHFFFAOYSA-N |
| SMILES | COc1cc2nc(C3CC3)nc(N3CCN(c4ccccc4OC)CC3)c2cc1OC |
| Storage condition | 2-8℃ |
Total 69, Current Page 1 of 7
1
2
3
4
5
| 2-Cyclopropyl-6,7-dimethoxy-4-[4-(2-methoxyphenyl)-1-piperazinyl]quinazoline |
| ML314 |