Agoniadin
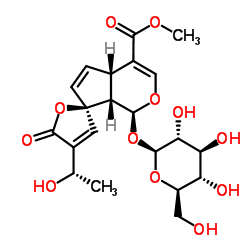
Agoniadin structure
|
Common Name | Agoniadin | ||
|---|---|---|---|---|
| CAS Number | 511-89-7 | Molecular Weight | 470.42 | |
| Density | 1.6±0.1 g/cm3 | Boiling Point | 771.4±60.0 °C at 760 mmHg | |
| Molecular Formula | C21H26O12 | Melting Point | 156-158°; mp 224-225° | |
| MSDS | Chinese USA | Flash Point | 269.2±26.4 °C | |
Use of AgoniadinPlumieride is an antifungal agent. Plumieride has strong fungitoxicity against some dermatophytes. Plumieride has little cytotoxic activity against the P388 leukaemia cell line with an IC50 of 85 μg/mL[1]. |
| Name | methyl (1S,4aS,7R,7aS)-4'-[(1S)-1-hydroxyethyl]-5'-oxo-1-[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxyspiro[4a,7a-dihydro-1H-cyclopenta[c]pyran-7,2'-furan]-4-carboxylate |
|---|---|
| Synonym | More Synonyms |
| Description | Plumieride is an antifungal agent. Plumieride has strong fungitoxicity against some dermatophytes. Plumieride has little cytotoxic activity against the P388 leukaemia cell line with an IC50 of 85 μg/mL[1]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.6±0.1 g/cm3 |
|---|---|
| Boiling Point | 771.4±60.0 °C at 760 mmHg |
| Melting Point | 156-158°; mp 224-225° |
| Molecular Formula | C21H26O12 |
| Molecular Weight | 470.42 |
| Flash Point | 269.2±26.4 °C |
| Exact Mass | 470.142426 |
| PSA | 181.44000 |
| LogP | -4.23 |
| Vapour Pressure | 0.0±6.0 mmHg at 25°C |
| Index of Refraction | 1.645 |
| InChIKey | AOPMSFXOYJXDNJ-IRFSQMTFSA-N |
| SMILES | COC(=O)C1=COC(OC2OC(CO)C(O)C(O)C2O)C2C1C=CC21C=C(C(C)O)C(=O)O1 |
| Hazard Codes | Xi |
|---|---|
| RIDADR | NONH for all modes of transport |
| Precursor 0 | |
|---|---|
| DownStream 1 | |
|
Assessment of the genotoxic and mutagenic properties of Himatanthus articulatus bark extracts used as phytotherapeutic drug in the Amazon.
J. Ethnopharmacol. 147(2) , 474-80, (2013) Himatanthus articulatus (Apocynaceae) is a plant native to the Amazon, popularly used to treat external ulcers, tumors, inflammations, cancer, syphilis and malaria.To investigate the in vivo genotoxic... |
|
|
Bioactive iridoids and a new lignan from Allamanda cathartica and Himatanthus fallax from the Suriname rainforest.
J. Nat. Prod. 60(12) , 1294-7, (1997) Bioassay-guided fractionation of the EtOAc extract of both Allamanda cathartica and Himatanthus fallax (Apocynaceae) using the Sc-7 yeast strain resulted in the isolation of the weakly cytotoxic isopl... |
|
|
Structural modifications of plumieride isolated from Plumeria bicolor and the effect of these modifications on in vitro anticancer activity.
J. Org. Chem. 69(19) , 6165-72, (2004) Plumieride was isolated as one of the major components from the biologically active methanolic extract of the bark of Plumeria bicolor (family Apocynaceae). For investigating the effect of substituent... |
|
Name: Primary qHTS assay for small molecule inhibitors of Inositol hexaphosphate kinase 1 (...
Source: NCGC
External Id: IP6K1-p1
|
|
Name: Inhibition of HIV1 RT
Source: ChEMBL
Target: Reverse transcriptase/RNaseH
External Id: CHEMBL1018093
|
|
Name: Cytotoxicity against human breast cancer cells
Source: ChEMBL
Target: NON-PROTEIN TARGET
External Id: CHEMBL1017530
|
|
Name: Cytotoxicity against human melanoma cells
Source: ChEMBL
Target: NON-PROTEIN TARGET
External Id: CHEMBL1016691
|
|
Name: Small-molecule inhibitors of ST2 (IL1RL1)
Source: 20881
Target: interleukin-1 receptor-like 1 isoform [homo sapiens]
External Id: ST2_IL33_Inhibitors_Primary_Screening_77700
|
|
Name: Cytotoxicity against human colon cancer cells
Source: ChEMBL
Target: NON-PROTEIN TARGET
External Id: CHEMBL1017532
|
|
Name: Cytotoxicity against human lung cancer cells
Source: ChEMBL
Target: NON-PROTEIN TARGET
External Id: CHEMBL1017531
|
|
Name: Cytotoxicity against human HT1080 cells
Source: ChEMBL
Target: HT-1080
External Id: CHEMBL1016690
|
| 1-(b-D-Glucopyranosyloxy)-4a,7a-dihydro-4'-(1-hydroxyethyl)-5'-oxospiro[cyclopenta[c]pyran-7(1H),2'(5'H)-furan]-4-carboxylic Acid Methyl Ester |
| Agoniadin |
| Methyl (1S,4aS,7R,7aS)-1-(β-D-glucopyranosyloxy)-4'-[(1S)-1-hydroxyethyl]-5'-oxo-4a,7a-dihydro-1H,5'H-spiro[cyclopenta[c]pyran-7,2'-furan]-4-carboxylate |
| Plumieride |
 CAS#:132586-69-7
CAS#:132586-69-7
