Modification of the actin interface of skeletal myosin subfragment-1 by treatment with dibromobimane.
D Mornet, K Ue, P Chaussepied, M F Morales
Index: Eur. J. Biochem. 159(3) , 555-61, (1986)
Full Text: HTML
Abstract
Recently, by treating the head portion of skeletal myosin subfragment-1 (S1) with the bifunctional agent dibromobimane, we introduced an intramolecular covalent cross-link which resulted in the stabilisation of an internal loop in the heavy chain structure of the head [Mornet et al. (1984) Proc. Natl Acad. Sci. USA 82, 1658-1662]. In order to define the functional properties of this new S1 conformational state, we have first determined the experimental conditions for the optimum modification of S1 by dibromobimane. We finally settled on a 60% yield of cross-linked S1. Because the modification occurs between the 50-kDa and the 20-kDa tryptic heavy chain fragments which have been postulated to be involved in the interaction of native S1 with actin, we have investigated the association of dibromobimane-treated S1 with actin, using chemical cross-linking of their rigor complex with 1-ethyl-3-[3-(dimethylamino)propyl] carbodiimide. The cross-linked species obtained were analyzed by polyacrylamide gel electrophoresis and compared with those known for unmodified S1. The carbodiimide-catalyzed linkage between actin and dibromobimane-modified S1 led to a singlet protein band migrating with an apparent molecular mass of 155 kDa, in contrast to the usual doublet bands of 175 kDa and 185 kDa produced with native S1. This result suggests that a change has occurred at the actin interface on the dibromobimane-treated S1 heavy chain. The covalent complex generated by carbodiimide cross-linking between actin and dibromobimane-modified S1 (27-kDa + 50-kDa + 20-kDa fragments) was submitted to chemical hydrolysis with hydroxylamine. The nature of the products identified is consistent with the conclusion that the internal freezing of the heavy chain structure by dibromobimane induces the loss of the ability to cross-linkage of the actin site on the 20-kDa domain but does not affect the conformation of the second site on the 50-kDa segment, which becomes the unique actin region cross-linkable by actin.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
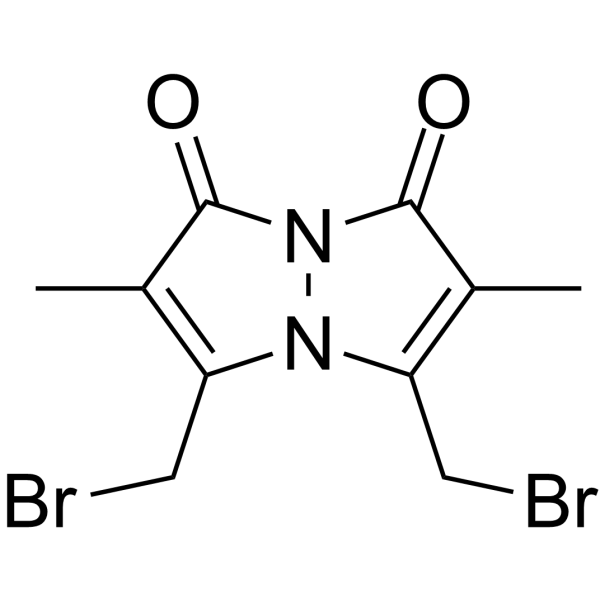 |
Dibromobimane
CAS:68654-25-1 |
C10H10Br2N2O2 |
|
Thiol labeling with bromobimanes.
1987-01-01 [Meth. Enzymol. 143 , 76, (1987)] |
|
Bromobimanes--fluorescent labeling agents for histochemical ...
1987-01-01 [Histochemistry 86 , 281, (1987)] |
|
Physicochemical changes in phosphorylase kinase induced by i...
2013-04-01 [Protein Sci. 22(4) , 444-54, (2013)] |
|
The C-terminus of H-Ras as a target for the covalent binding...
2011-01-01 [PLoS ONE 6(1) , e15866, (2011)] |
|
ArsD residues Cys12, Cys13, and Cys18 form an As(III)-bindin...
2007-06-08 [J. Biol. Chem. 282(23) , 16783-91, (2007)] |