Mechanism of inactivation of rat liver microsomal cytochrome P-450c by 2-bromo-4'-nitroacetophenone.
A Parkinson, P E Thomas, D E Ryan, L D Gorsky, J E Shively, J M Sayer, D M Jerina, W Levin
Index: J. Biol. Chem. 261(25) , 11487-95, (1986)
Full Text: HTML
Abstract
The mechanism by which 2-bromo-4'-nitroacetophenone (BrNAP) inactivates cytochrome P-450c, which involves alkylation primarily at Cys-292, is shown in the present study to involve an uncoupling of NADPH utilization and oxygen consumption from product formation. Alkylation of cytochrome P-450c with BrNAP markedly stimulated (approximately 30-fold) its rate of anaerobic reduction by NADPH-cytochrome P-450 reductase, as determined by stopped flow spectroscopy. This marked stimulation in reduction rate is highly unusual in that Cys-292 is apparently not part of the heme- or substrate-binding site, and its alkylation by BrNAP does not cause a low spin to high spin state transition in cytochrome P-450c. Under aerobic conditions the rapid oxidation of NADPH catalyzed by alkylated cytochrome P-450c was associated with rapid reduction of molecular oxygen to hydrogen peroxide via superoxide anion. The intermediacy of superoxide anion, formed by the one-electron reduction of molecular oxygen, established that alkylation of cytochrome P-450c with BrNAP uncouples the catalytic cycle prior to introduction of the second electron. The generation of superoxide anion by decomposition of the Fe2+ X O2 complex was consistent with the observations that, in contrast to native cytochrome P-450c, alkylated cytochrome P-450c failed to form a 430 nm absorbing chromophore during the metabolism of 7-ethoxycoumarin. Alkylation of cytochrome P-450c with BrNAP did not completely uncouple the catalytic cycle such that 5-20% of the catalytic activity remained for the alkylated cytochrome compared to the native protein depending on the substrate assayed. The uncoupling effect was, however, highly specific for cytochrome P-450c. Alkylation of nine other rat liver microsomal cytochrome P-450 isozymes with BrNAP caused little or no increase in hydrogen peroxide formation in the presence of NADPH-cytochrome P-450 reductase and NADPH.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
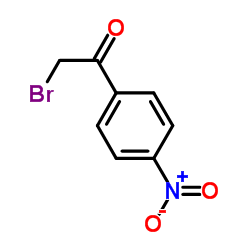 |
p-nitrophenacyl bromide
CAS:99-81-0 |
C8H6BrNO3 |
|
QSAR study and conformational analysis of 4-arylthiazolylhyd...
2015-10-12 [Eur. J. Pharm. Sci. 78 , 190-7, (2015)] |
|
Active site of bee venom phospholipase A2: the role of histi...
1996-04-09 [Biochemistry 35(14) , 4591-601, (1996)] |
|
Chemical modification and inactivation of rat liver microsom...
1986-09-05 [J. Biol. Chem. 261(25) , 11478-86, (1986)] |
|
[Amphoteric ion intermediates in the pyrazine series. II. Th...
1990-01-01 [Rev. Med. Chir. Soc. Med. Nat. Iasi. 94(1) , 157-60, (1990)] |
|
Use of an imperfect neutral diluent and outer vesicle layer ...
1997-04-01 [Biochemistry 36(13) , 3870-81, (1997)] |