Toward chemistry-based design of the simplest metalloenzyme-like catalyst that works efficiently in water.
Taku Kitanosono, Shū Kobayashi
Index: Chem. Asian J. 10(1) , 133-138, (2015)
Full Text: HTML
Abstract
Enzymes exhibit overwhelmingly superior catalysis compared with artificial catalysts. Current strategies to rival enzymatic catalysis require unmodified or minimally modified structures of active sites, gigantic molecular weight, and sometimes the use of harsh conditions such as extremely low temperatures in organic solvents. Herein, we describe a design of small molecules that act as the simplest metalloenzyme-like catalysts that can function in water, without mimicking enzyme structures. These artificial catalysts efficiently promoted enantioselective direct-type aldol reactions using aqueous formaldehyde. The reactions followed Michaelis-Menten kinetics, and heat-resistant asymmetric environments were constructed in water. © 2015 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
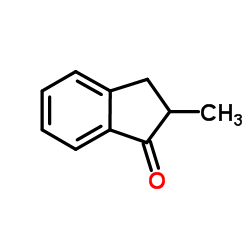 |
2-Methyl-indanone
CAS:17496-14-9 |
C10H10O |
|
The O-acylation of ketone enolates by allyl 1H-imidazole-1-c...
2007-11-23 [J. Org. Chem. 72(24) , 9372-5, (2007)] |
|
Recent advances in enzymatic and chemical deracemisation of ...
2013-12-21 [Chem. Soc. Rev. 42(24) , 9268-9282, (2013)] |
|
Amino alcohol-mediated enantioselective syntheses of α-...
[Tetrahedron Asymmetry 25(9) , 697-704, (2014)] |
|
Structural effects in the Pd-induced enantioselective deprot...
[Tetrahedron Asymmetry 18(24) , 2859-2868, (2007)] |
|
Heck-type reactions of allylic alcohols: Part IV:(2-Substitu...
[J. Mol. Catal. A: Chem. 283(1) , 140-45, (2008)] |