| Structure | Name/CAS No. | Articles |
|---|---|---|
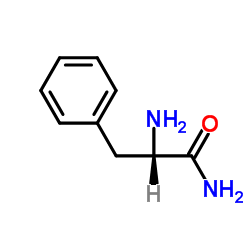 |
H-Phe-NH2
CAS:5241-58-7 |
|
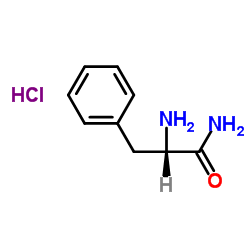 |
L-Phenylalanin amide hydrochloride
CAS:65864-22-4 |