Alpha1A-adrenergic receptors are functionally expressed by a subpopulation of cornu ammonis 1 interneurons in rat hippocampus.
Kristin L Hillman, Van A Doze, James E Porter
Index: J. Pharmacol. Exp. Ther. 321(3) , 1062-8, (2007)
Full Text: HTML
Abstract
The importance of adrenergic receptors (ARs) in the hippocampus has generally focused on betaARs; however, interest is growing in hippocampal alphaARs given their purported neuroprotective role. We have previously reported alpha(1)AR transcripts in a subpopulation of cornu ammonis 1 (CA1) interneurons. The goal of this study was to identify the specific alpha(1)AR subtype (alpha(1A), alpha(1B), alpha(1D)) functionally expressed by these cells. Using cell-attached recordings to measure action potential frequency changes, concentration-response curves for the selective alpha(1)AR agonist phenylephrine (PE) were generated in the presence of competitive subtype-selective alpha(1)AR antagonists. Schild regression analysis was then used to estimate equilibrium dissociation constants (K(b)) for each receptor antagonist in our system. The selective alpha(1A)AR antagonists, 5-methylurapidil and WB-4101 [2-[(2,6-dimethoxyphenoxyethyl)aminomethyl]-1,4-benzodioxane hydrochloride], produced consecutive rightward shifts in the concentration-response curve for PE when used at discriminating, nanomolar concentrations. Calculated K(b) values for 5-methylurapidil (10 nM) and WB-4101 (5 nM) correlate to previously published affinity values for these antagonists at the alpha(1A)AR. The selective alpha(1B)AR antagonist L-765,314 [(2S)-4-(4-amino-6,7-dimethoxy-2-quinazolinyl)-2-[[(1,1-dimethylethyl)amino]carbonyl]-1-piperazinecarboxylic acid], as well as the selective alpha(1D)AR antagonist BMY7378 [8-[2-[4-(2-methoxyphenyl)-1-piperazinyl]ethyl]-8-azaspiro[4.5]decane-7,9-dione dihydrochloride], produced significant rightward shifts in the concentration-response curve for PE only when used at nondistinguishing, micromolar concentrations. Calculated K(b) values for L-765,314 (794 nM) and BMY7378 (316 nM) do not agree with affinity values for these antagonists at the alpha(1B) or alpha(1D)AR, respectively. Rather, these K(b) values more closely match equilibrium dissociation constants estimated for these compounds when used to identify alpha(1A)AR subtypes. Together, our results provide strong evidence to support functional expression of alpha(1A)ARs in a subpopulation of CA1 interneurons.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
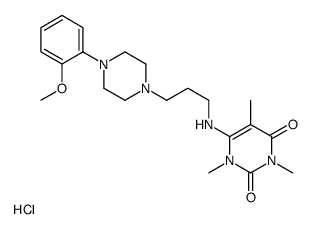 |
5-Methylurapidil
CAS:34661-85-3 |
C21H32ClN5O3 |
|
Cell membrane chromatography competitive binding analysis fo...
2011-07-01 [Anal. Bioanal. Chem 400(10) , 3625-33, (2011)] |
|
Influence of high dietary sodium intake on the functional su...
2009-01-01 [Auton. Autacoid Pharmacol. 29(1-2) , 25-31, (2009)] |
|
Alpha-1A adrenergic receptor activation increases inhibitory...
2009-04-01 [Epilepsy Res. 84(2-3) , 97-109, (2009)] |
|
Pharmacological characterization of alpha1- and beta-adrener...
2006-02-01 [Arch. Physiol. Biochem. 112(1) , 23-30, (2006)] |
|
Influence of cisplatin-induced renal failure on the alpha(1)...
2007-08-13 [Eur. J. Pharmacol. 569(1-2) , 110-8, (2007)] |